Epigenetics: How the environment influences our genes
PDF
Our environment influences our genome through the so-called epigenetic modifications. For example, with identical genetic heritages, two twins may evolve differently depending on their respective environments. There is growing evidence that our genes “listen” to the environment in complex ways that affect our health and behaviour.The diets, many iterative diseases, the drugs and the toxins, anxiety and stress, our lifestyle & where we live can modify both the host cells and their DNA. Thus, in monozygotic twin pairs, large differences in life trajectory could be found. One could develop obesity and the other could remain thin; one could be sane and the other could develop a mental illness. A few examples from animals and humans highlight the role of epigenetics [1] in the transmission of acquired traits.
1. Methylation: the keystone of epigenetics
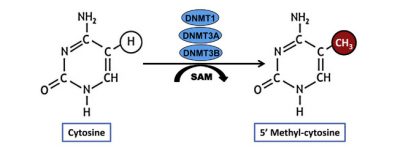
It is now known that genes can be “turned on” or “off” by several types of chemical modifications that do not change the DNA sequence: DNA methylations and histone modifications, those proteins on which DNA is wrapped to form chromatin [2]. All these modifications constitute “epigenetic marks” [3] grouped under the term epigenome (Figures 1 & 2). Epigenetic modifications are induced by the environment in the broad sense: the cell constantly receives all kinds of signals informing it about its environment, so that it specializes during development, or adjusts its activity to the situation (see Epigenetics, the Genome and its Environment). These signals, including those related to our behaviours (diet, smoking, stress, etc.), can lead to changes in the expression of our genes, without affecting their sequence. The phenomenon may be transient, but there are permanent epigenetic changes, which persist when the signal that induced them disappears. Unlike genetic mutations, which are irreversible, epigenetic “tagging” can change. A simple change of environment can change the way the genes we inherit at birth, and therefore our “phenotype” [4].
In addition, there is another scale in epigenetic processes since there is a set of regulatory elements of the epigenetic processes themselves. Either regulatory structures inserted into the DNA sequence, sometimes at great distance from genes, or mobile small molecules called microRNAs (miRNAs) or interfering small RNAs are involved in many processes in eukaryotes (e.g. methylation elimination, chromatin assembly, and suppression of protein formation that are activated in immediate stress responses). Much smaller than messenger RNAs, these miRNAs prevent them from conveying their message, by neutralizing or destroying them.
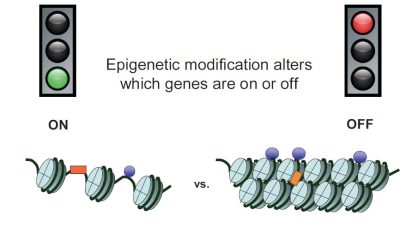
In other words, if the chromosome is the magnetic tape of a cassette and each gene corresponds to a track recorded on the tape, epigenetic modifications are pieces of repositionable adhesive tape that will mask or unmask certain tracks, making them unreadable or readable. Regarding the modifications that mask, we speak about methylating agents, responsible for the methylation of DNA and histones (see above). On the other hand, other molecules have the effect of stimulating gene expression. These are called “acetyl groups” and are referred to as acetylating agents and histone acetylation.
2. What are the consequences for the animal kingdom?
2.1. General information
Some epigenetic marks are likely to pass to offspring. The intergenerational transmission of marks materialized by DNA methylation is well documented in plants. In mammals, the study of the phenomenon is much more complex and still controversial. In this paragraph and through some examples taken from animals (the Agouti mice, Drosophila red eyes and cross-adoption experiments in rats), we observe epigenetics as a field studying the influence of the environment and individual history on gene expression.
2.2. The Agouti mouse model

It was not until the end of the 20th century that a type of mouse was discovered allowing scientists to highlight the complex connections linking food to epigenetics. Among the many genes that contribute to coat colour in mice, one of them is called “Agouti”. The most interesting version of the Agouti gene is known as “Agouti viable yellow” or Avy (Figure 3). If the Avy gene has little or no methylation, it is active in all cells, and mice are yellow. These yellow mice are susceptible to the development of diseases such as obesity, diabetes or certain cancers. But if Avy is hypermethylated, his expression “fades out”, which implies that the mouse has a brown color, and has no health problems, even if it has exactly the same Agouti gene as yellow mice. Between these two extremes, Avy can be methylated to varying degrees, which affects the level of activity of the gene [5]. The result is a beautiful gradation of spotted mice, in which the activity of the Avy gene even differs from one cell to another. The same genetically identical litter of mice varies in colour according to this spectrum, due to epigenetic variations established in the uterus. In addition, regardless of coat colour, it highlights the effects of diet on methylation. Randy Jirtle, an American researcher, has had a remarkable experience with these mice carrying the Agouti gene. By feeding them B vitamins, he did not “cure” these genetically ill mice, but the beneficial effect was felt on the offspring [6]. In other words, the offspring of mice carrying the Agouti gene fed with B vitamins are no longer sick or even beige (the Agouti gene is still there, but it is no longer expressed), while the offspring of those who have not received B vitamins remain sick from generation to generation!
2.3. Drosophila and red eyes

Drosophila are insects commonly used in the laboratory. Their genome is relatively simple to understand and as a result, they are the “stars” of genetic research… In April 2009, Dr Renato Paro, from the University of Basel, announced a wonderful new discovery concerning them: if a fruit fly egg is heated to 37° degrees before hatching, the fly has red eyes. Otherwise, her eyes are white….. Better! The “red eye” character has been passed down from generation to generation. It is therefore a characteristic acquired through the influence of an external factor (temperature) that becomes hereditary [7]. These animal studies, like the following studies, seem to support the scientific theory represented by Lamarckism. According to his famous publication – “The Influence of Circumstances” – published in 1809-1810 [8], Lamarck supports the idea that physical changes acquired during an individual’s lifetime could be transmitted to his descendants [9]. However, in humans we have no evidence of the persistence of these epigenetic effects beyond a few generations. These results do not therefore really call into question the Darwinian conception of the long-term evolution of species through the action of natural selection on accidental hereditary variations (see Theory of evolution: misunderstandings and resistance & Adaptation: responding to environmental challenges).
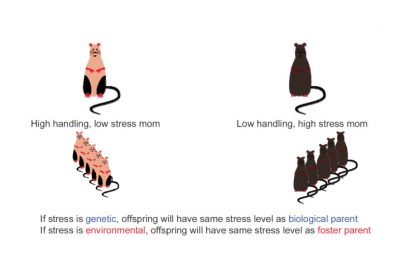
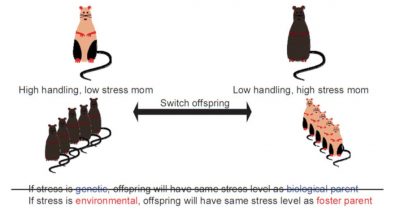
2.4. Mouse mothering: beyond the uterus
Do simple caresses also have the power to influence genes? In rats, licking fulfils the same function as caressing in humans. However, studies show that baby rats often licked by their mothers are calmer. But at Mc Gill University in Montreal (Canada), Professor Michael Meaney’s team went much further by revealing footprints of this maternal care into the brains of young rats, at the hippocampus level [10].
In fact, it is the “licking” that influences the activity of a gene that protects rats against stress. This gene, called NRC31, produces a protein (glucocorticoid receptor GC) that helps to reduce the concentration of stress hormones (cortisol) in the body [11]. However, a specific portion of this gene must be activated using the epigenetic switch represented by DNA methylation. Analysis of the brains of rats that did not receive enough affection by licking demonstrated this: the switch linked to the NRC31 gene (gene coding for the glucocorticoid receptor) was defective (gene that had undergone methylation, and therefore inactive) in the neurons of the rats hippocampus (a brain area that incorporates ambient stresses). As a result, the amount of stress hormones (cortisol) increases in the blood and therefore, even in the absence of disruptive elements, they live in a state of constant stress.
3. Impact of food on our genes

In order to show the link between epigenetics and diet, especially in humans, let’s go back to the history of the dramatic “Dutch hunger famine of 1944“. It offers scientists a kind of life-size laboratory to test these hypotheses. During the winter of 1944-1945, the west of the Netherlands was starving due to a blockade by Nazi Germany [12].
Studies have shown that children of pregnant women exposed to this famine got pathologies such as diabetes, obesity, cardiovascular disease, microalbuminuria (albumin in the urine), etc. In addition, they were smaller than normal. As adults, they then had smaller than average children too! [13].
These data suggest that the famine experienced by mothers caused epigenetic changes that were passed on to subsequent generations. Their genetic heritage had kept track of this event, which supports the theory that an individual’s health is partly programmed by the environment to which the foetus is exposed in the uterus. We now know that these effects on size come from changes in the epigenetic markers present on their DNA, linked to deficiencies in certain crucial molecules in the diet of Dutch grandmothers during this period. Indeed, in order to faithfully maintain a good level of methylation during cell divisions, new methyl groups must be added to the newly copied DNA. However, this constant supply of methyl groups comes directly from our food from amino acids and vitamins (such as methionine, betaine or choline). In addition, we can also manufacture “methyl” groups from chemical precursors such as folic acid.
But in 2008, researchers further elucidated these epigenetic changes [14] : individuals who have experienced famine in utero have fewer methyl groups attached to the gene that controls the production of a growth factor, IGF-2 (insulin-likegrowth factor-2). Other chemical elements from our food are also needed to transport methyl groups through the body and carefully attach them to DNA: zinc, vitamin B12…
4. What lessons can be learned from this?
While the genome is fixed, the epigenome is much more dynamic. Epigenetic modifications would allow individuals to quickly explore an adaptation to a change in the environment, without “engraving” this adaptive change into the genome. The challenges of epigenetics concern not only medicine and public health (see Epigenetics, the Genome and its Environment) but also evolution (see Theory of evolution: misunderstandings and resistance). Indeed, it casts suspicion on the environment that could modulate the activity of some of our genes to modify our traits, or even induce certain diseases potentially transmissible to offspring. Clearly, the Dutch famine of the winter of 1944-1945 shows that permanent changes have occurred in the genetic heritage of the women who were pregnant at this time and then passed on from generation to generation. This would mean that the trauma also affects the germ cells (sperm and eggs), the only biological link between generations.
It is now widely accepted that epigenetic abnormalities contribute to the development and progression of human diseases, in particular cancers. Epigenetic processes are involved in the regulation of many events such as cell division, differentiation (specialization of cells in a particular role), survival, mobility… The alteration of these mechanisms promotes the transformation of healthy cells into cancer cells, any epigenetic aberration can be involved in carcinogenesis. Epigenetic abnormalities activating oncogenes (genes which overexpression promotes carcinogenesis) or inhibiting tumor suppressor genes have been identified. Similarly, mutations affecting genes encoding the enzymes responsible for epigenetic markings have been identified in tumour cells. It remains to be seen whether these phenomena are the cause or consequence of cancer development. Nevertheless, it seems that they participate in tumor progression (or cancer progression).
In addition, the role of epigenetics is suspected and extensively studied in the development and progression of complex and multifactorial diseases, such as neurodegenerative diseases (Alzheimer’s, Parkinson’s, amyotrophic lateral sclerosis, Huntington…) or metabolic diseases (obesity, type 2 diabetes…). In the same way that we now know how to obtain the sequence of a complete genome, it is also possible to know all the epigenetic modifications that characterize it: we speak of an epigenome. It is this type of global and unbiased approach that will make it possible to better understand the involvement of epigenetics in human diseases…
What about telomerases?
In addition to the epigenetic processes discussed more than 50 years ago by the British biologist Conrad Waddington (1905-1975) [15] and mentioned above, we must mention a phenomenon of crucial importance, and sensitive to our environment. This is a major discovery in molecular biology initiated by Elizabeth Blackburn and Carol Greider, the laureates of the 2009 Medicine & Physiology Nobel Prize: the identification of an enzyme called telomerase. This enzyme regulates the length ofthe “ telomers”, which are repeated segments of non-coding DNA, located at the end of each chromosome (see focus Slowing down aging: the telomerase track?). These telomeres can be represented as the small plastic pieces that protect the tips of your shoelaces and are called “aglets”. These telomers form small caps at the ends of the chromosomes, preventing the genetic material from “fraying”. These are, among other things, the aglets of aging and they tend to shorten over time. Thus, under the influence of telomerase, telomeres can stop shortening and even lengthen. Aging is therefore a dynamic process that can be accelerated or slowed down. The work of Elizabeth Blackburn and nutritionist medical doctor Dean Ornish has clearly shown the effects of lifestyle changes on the lengthening of these telomeres (see focus Slowing down aging: the telomerase track?) [16].
In conclusion, the importance of the role of the environment in epigenetic heredity is far from being resolved, despite the announcement effects that have led to a renewed interest in Lamarck’s thesis of “the heritability of acquired traits” (see Lamarck and Darwin: two divergent visions of the living world). The ultimate inquiry is that of the importance of epigenetic processes in Evolution. The scientific community remains divided and a central question remains: are epigenetic states transmitted over a sufficient number of generations to give rise to natural selection? Do doctors and sociologists ask another one, which interests us in the short and medium term: Does our lifestyle matter more than our heredity?
References and notes
Cover image. These twins have the same genotype, but their epigenotype and phenotype are different because they did not live in the exact same environment. [Source : Photo © Peter Voerman photography via Visual Hunt / CC BY-NC.]
[1] Epigenetics: All changes in gene activity that are transmitted over cell divisions or over generations without the use of DNA mutations. This “memory of gene activity” is based on chromatin states, or “epigenetic marks”.
[2] Chromatin: A basic substance of chromosomes consisting of the DNA molecule associated with proteins called “histones”, around which it is wrapped.
[3] Epigenetic marks: Chemical modifications of DNA or associated proteins in chromatin. These modifications (e.g. cytosine methylation or C) contribute to the control of gene activity without affecting the nucleotide sequence, the “letters” (A, T, C or G) that write the gene message on the DNA.
[4] Jablonka E & Lamb M (2005) Evolution in four dimensions. Cambridge MA. MIT press
[5] Morgan HD et al. (1999) Epigenetic Inheritance at the agouti locus in the mouse. Nature genetics 23: 314-318
[6] Jirtle, RL & Tyson, FL, eds. (2013) Environmental Epigenomics in Health and Disease: Epigenetics and Complex Diseases Origins, Heidelberg: Springer
[7] G & R Paro Shit (2009) About Combs, Notches, and Tumors: Epigenetics Meets Signaling. Developmental Cell 17: 440-442
[8] Lamarck JB (1809-1810) Influence des circonstances sur les actions des animaux (5e leçon), Muséum National d’Histoire Naturelle de Paris (in french)
[9] Evolution according to Charles Darwin (1809-1882): Species evolve as a result of “variations” (character changes) that occur randomly, and are transmitted to descendants. Only the advantageous (“adaptive”) variations are selected by the environment: this is “natural selection”. Evolution according to Jean-Baptiste de Lamarck (1744-1829): The environment would dictate a beneficial change for the individual, which would be transmitted to his descendants. This “heredity of acquired traits” would govern the evolution of species.
[10] Francis D et al (1999) Nongenomic transmission across generations of Maternal behavior and stress in the rat. Science 1155-1158
[11] Weaver IC et al (2004) Epigenetic programming by maternal behavior. Nature Neuroscience 7: 847-854
[12] Lumey LH et al (2007) Cohort profile: The dutch Hunger Winter Families Study. Int J Epidemiol. 36: 1196-1204
[13] Stein AD et al (2004) Intrauterine famine exposure and body proportions at birth: the Dutch Hunger Winter. Int J Epidemiol 33: 831-836
[14] Heijmans BT et al (2008) Persistent epigenetic differences associated with prenatal exposure to famine in humans. PNAS 17046-17049
[15] Baedke J (2013) The epigenetic landscape in the course of time: Conrad Hal Waddington’s methodological impact on the life sciences. Studies in History and Philosophy of Science Part C: Studies in History and Philosophy of Biological and Biomedical Sciences 44 : 4 Part B 756-773
[16] Ornish D et al. (2008) Increased telomerase activity and comprehensive lifestyle changes: a pilot study. Lancet Oncol. 2008 9(11):1048-57
The Encyclopedia of the Environment by the Association des Encyclopédies de l'Environnement et de l'Énergie (www.a3e.fr), contractually linked to the University of Grenoble Alpes and Grenoble INP, and sponsored by the French Academy of Sciences.
To cite this article: DROUET Emmanuel (September 6, 2021), Epigenetics: How the environment influences our genes, Encyclopedia of the Environment, Accessed April 18, 2024 [online ISSN 2555-0950] url : https://www.encyclopedie-environnement.org/en/health/epigenetics-how-the-environment-influences-our-genes/.
The articles in the Encyclopedia of the Environment are made available under the terms of the Creative Commons BY-NC-SA license, which authorizes reproduction subject to: citing the source, not making commercial use of them, sharing identical initial conditions, reproducing at each reuse or distribution the mention of this Creative Commons BY-NC-SA license.







