The first complex ecosystems
PDF
Today’s ecosystems are amazing in the complexity of the interdependent relationships between the organisms that make up them. Energy and biomass circulate through food webs that unite bacteria, unicellular organisms, plants/algae and animals of extremely varied nature and size. The stability of these biological systems is based on dynamic balances that are nevertheless very sensitive to environmental and anthropogenic factors. For more than three billion years, marine ecosystems have been dominated by microbial organisms (bacteria, archaea) or unicellular eukaryotes. While these organisms have played a key role in the biogeochemical cycles of carbon or nitrogen and in raising the level of oxygen on our planet, they have never formed complex food webs as we know them in nature today. A little over 500 million years ago, the appearance of multicellular and macroscopic organisms at the end of the Precambrian and the advent of the animal kingdom during the Precambrian-Paleozoic transition revolutionized the marine world and its mode of operation.
1. The enigmatic marine ecosystem of the Ediacaran

Present in many fossil sites in Canada (e.g. Mistaken Point), Australia (e.g. Ediacara), Namibia and northern Russia (e.g. White Sea), these enigmatic organisms have colonized the seabed in abundance at variable depths between 575 and 542 million years, a geological interval corresponding to the end of the Ediacaran. The footprints of their soft, fleecy and flexible bodies, without equivalent in today’s nature, have been preserved thanks to the instantaneous deposition of sandy sediments or volcanic ash.
Among the most typical, rangeomorphs [1] are characterized by a wavy frondoStructure of a flattened, relatively large, leaf-like living organ or organism. with a stem firmly anchored to the bottom. These fixed organisms, unique in their modular and fractal structure, do not, however, reach the anatomical complexity of the first Cambrian animals. Apparently devoid of mouth, digestive system and complex internal organs, they were thought to have extracted their food by direct absorption of dissolved organic carbon (osmotrophyA mode of feeding that consists of feeding from dissolved substances. The osmotrophic organisms are nourished by transmembrane exchange, i.e. by diffusion of ions or small molecules through the cytoplasmic membrane. This type of nutrition, which is very common among microorganisms, is also provided by a number of animals, both free and parasitic. It is only possible in liquid environments (aquatic environments, internal fluids of animals or plants) or by the synthesis of enzymes that “digest” their solid environment.) thanks to their very large surface of exchange with the environment.
At the Ediacaran, the water-sediment interface of the ocean floor was also occupied by sponges and many flattened organisms sometimes evoking the bilateral symmetry of some molluscsEmbranchement of invertebrate animals, not segmented, with bilateral symmetry sometimes altered. They have a soft body (hence the name mollusk) usually composed of a head, a visceral mass, and a foot. They may have a calcareous shell produced by a mantle covering the visceral mass. and current arthropodsBranch of invertebrate animals whose organisational plane is characterised by a body segmented with articulated appendages and covered by a rigid cuticle or shell, which constitutes their exoskeleton, in most cases composed of chitin. The arthropod branch appeared 543 million years ago and is by far the one with the most species and individuals in the entire animal kingdom (80% of known species). (Figure 1). Traces produced by some of them such as Kimberella, Dickinsonia and Yorgia indicate that they moved and consumed the bacterial filmsAlso called biofilms; microbial community marked by the secretion of an adhesive and protective matrix. It is usually formed in water or in an aqueous medium. Biofilms were probably the first colonies of living organisms more than 3.5 billion years ago. With stromatolites, they seem to be the origin of the first biogenic rocks and reef structures, which then covered the entire seabed, probably by external digestion along their ventral surface as in the placozoairesMetazoans (animals) with the simplest organizational plan. These tiny (between 1 and 3 mm) flattened animals have no symmetry, no mouth, no digestive tract, no nervous system, no basal blade. They have no organs and only four different types of somatic cells. The vast majority of Ediacaran organisms appear to belong to evolutionary lines that appeared before those of animals in the strict sense ({tooltip}Eumetazoaires{end-text}Higher (animal) metazoans including all major animal groups except sponges and placozoa.).
The marine ecosystem of that time was essentially dominated by microbial mats and osmotrophic multicellular organisms (e. g. rangeomorphs), microphagesAnimals consuming very small amounts of solid food (particles) that must be absorbed in large quantities. The particles ingested range from organic debris a few nanometres in size to shellfish and shrimp. This is an important part of the krill on which whales feed (e.g. sponges via their filter system and their flagellatedcharacteristics of unicellular cells or organisms equipped with one or more flagella, structure ensuring their mobility. cells) or using external contact digestion. These feeding strategies were perfectly adapted to the resources available in the marine environment at the end of the Precambrian, namely an abundant flow of dissolved organic matter and microbial mats ubiquitous at the water-sediment interface. The extinction of Ediacaran organisms at the Precambrian-Cambrian transition would not be due to a global environmental upheaval like most major biological crises, but rather to the destruction of their biotopeLocation with relatively uniform determined physical and chemical characteristics. This environment is home to a set of life forms that make up biocenosis: flora, fauna, micro-organisms. A biotope and the biocenosis it supports form an ecosystem. by the very first burrowing animals (bioturbationActive mixing of soil or water layers by living species, mainly animals.). Completely defenceless, these organisms would not have survived the first predators.
2. The first animal communities: prototype of modern ecosystems
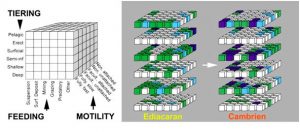
By definition, Cambrian Explosion refers to the relatively sudden appearance in the fossil record of both new and anatomically complex organisms, among which we can certainly recognize the distant ancestors of the main current animal groups (e. g. arthropods, worms, molluscs, chordates; Figure 2). Several exceptionally preserved deposits (called LagerstättenGerman word, plural, literally meaning “storage area”. Corresponds to geological sites of extreme fossil richness that have been remarkably preserved () such as Chengjiang (China; approx. 520 Ma), Burgess Shales (Canada; approx. 505 Ma), Sirius Passet (Greenland) and Emu Bay (Australia) reveal the existence of the first marine animal communities. Thanks to already developed motor and sensory capacities (e.g., sometimes fossilized cephalic nervous system), these early Cambrian animals were able to move actively in their environment and exploit for the first time a multitude of ecological niches. This dynamic marks a fundamental difference with the essentially fixed marine life of the Ediacaran and an irreversible turning point in the evolution of ecosystems.
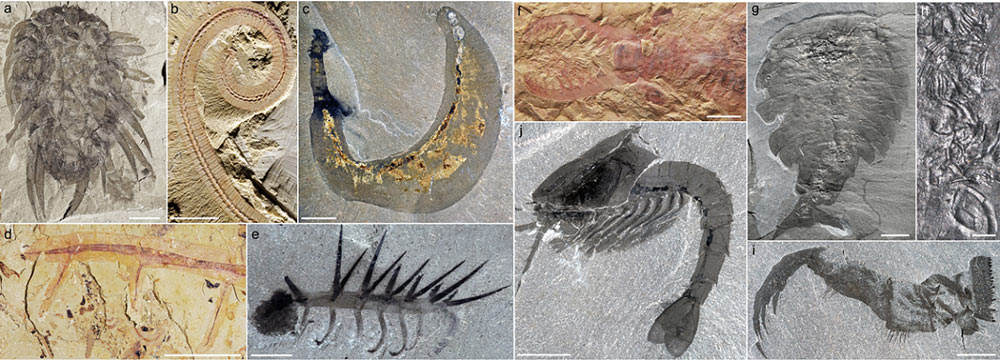


– cnidariansA branch (phylum) of aquatic animals (mainly marine) that is found in two forms: polyps, when fixed (as in the case of coral or sea anemones), and jellyfish when they are swimmers.,
– ctenophoresSmall, hermaphroditic, predatory marine organisms. They have a vague similarity to jellyfish and are a very important part of the plankton, – {tooltip}chaetognates{ind-text}A branch (phylum) of arrow-shaped marine predators named after the mobile hooks that capture their prey. They play a major role in the planktonic ecosystem as the main direct predators of copepods and represent up to 10% of the zooplankton biomass.,
– primary molluscs and arthropods (e.g. IsoxysExtinct type of small primary arthropods that lived in the Lower Cambrian. Their main characteristic is the existence of a pointed bivalve shell.).
The comparative study of these fossils and their current descendants (Figure 4) suggests that the species interacted within a primitive food chain [3],[4],[5],[6],[7],[8],[9],[10],[11]. The Lower Cambrian Timisiocari species of Greenland is a good example of these new trophic relationships. Close cousin of the emblematic predator Anomalocaris, its large appendages were equipped with combs and filter bristles allowing it to catch zooplanktonAnimal plankton. It feeds on living matter, some species being herbivores and others carnivores. living in suspension in the water column. Microfossils attest to the presence of this zooplankton that consumes eukaryotic algae and bacteria.
However, Cambrian marine life is concentrated at the water-sediment interface, with sponges representing a major component of sessile faunaOrganizations living alone or in colonies and permanently fixed directly to the substratum. They are most often aquatic. This is the case, for example, for sponges, corals, hydrozoa, tunicates, bryozoans, etc.. Arthropods are by far the most abundant and diverse epibenthic organismsOrganisms living on the surface of the bedrock in the seabed area. in all deposits with exceptional preservation (Figure 2). Their external (segments, appendages) and internal (nervous system) organization plan suggests, for some of them, kinship relationships with current crustaceans and cheliceratesGroup of arthropods carrying chelicera, a pair of appendages close to the mouth, corresponding to the second pair of antennas in mandibules (crustaceans, insects…). This group includes merostomes (limules) and arachnids (spiders, scorpions, etc.). Only the horseshoe crabs are marine animals and live on the bottom..
Others belong to groups that are now extinct. Their articulated and multi-segmented exoskeleton has probably favoured the acquisition of many functionalities and specializations. Prehensil and chewing appendages allow Cambrian arthropods to capture prey and reduce food particles. Macrophagia appears in Cambrian times in many predatory animals or animals that feed on corpses (scavengers). For example, the Sidneyia arthropod of the Burgess Shale captured, crushed and consumed small trilobitesA class of fossil marine arthropods that existed during the Paleozoic (primary era) from Cambrian to Permian. The last trilobites disappeared during the mass extinction at the end of the Permian, there are 250 Ma. as indicated by its appendages and stomach contents (Figure 2).
Other innovations contribute to the major changes in the food chain. Digestive glands increase the efficiency of enzymatic degradation of food, thus promoting macrophagiaMode of nutrition of a living organism that feeds on large prey compared to it. in many arthropods in Cambodia. The vision also revolutionized the interactions between marine organisms from the early Cambrian period. Thus, large compound eyes made up of thousands of facets allowed the super-predator Anomalocaris to spot and track its prey. There is no doubt that vision, widespread among Cambrian arthropods, has significantly altered prey-predator relationships and introduced new selection pressures within the ecosystem, leading to multiple adaptive responses.
References and notes
Cover image. Life in the Ediacara Sea [© Ryan Somma (CC BY-SA 2.0), via Wikimedia Commons]
[1] Narbonne G.M., Laflamme M., Greentre C. Trusler P. (2009) Reconstructing a lost world: Ediacaran rangeomorphs from Spaniard’s Bay, Newfoundland. Journal of Paleontology 83, 503-528.
[2] Fedonkin M.A., Gehling J.G., Grey K., Narbonne G. Vickers-Rich P. (2007) The Rise of Animals. The Johns Hopkins University Press, Baltimore. 325 pp.
[3] Briggs D.E.G. (2015) The Cambrian Explosion. Current Biology 25, R864-R868
[4] Caron J.-B., Scheltema A., Schander C. & Rudkin D. (2006) A soft-bodied mollusc with radula from the Middle Cambrian Burgess Shale. Nature 442, 159-163.
[5] Caron J.-B., Conway Morris S. & Cameron C.B. (2013) Tubicolous enteropneusts from the Cambrian period. Nature 495, 503-506.
[6] Daley et al (2009) The Burgess Shale anomalocarididid Hurdia and its significance for early euarthropod evolution. Science 323, 1597-1600.
[7] Hou X.-G., Aldridge R.J., Bergström J., Siveter David J., Siveter Derek J. & Feng X.-H. (2004) The Cambrian Fossils of Chengjiang, China. Blackwell Publishing. 233 pp.
[8] Smith M.R. & Caron J.-B. (2010) Primitive soft-bodied cephalopods from the Cambrian. Nature 465, 469-472.
[9] Vannier J., Steiner M., Renvoisé E., Hu S.-X. Casanova J.-P. (2007) Early Cambrian origin of modern food webs: evidence from predator arrow worms. Proceedings of the Royal Society London B 274, 627-633.
[10] Vannier J., Garcia-Bellido D.C., Hu S.-X. Chen A.L. (2009) Arthropod visual predators in the early pelagic ecosystems: evidence from the Burgess Shale and Chengjiang biota. Proceedings of the Royal Society London B 276, 2567-2574.
[11] Vinther J., Stein M., Longrich N.R. & Harper D.A.T. (2014) A suspension-feeding anomalocarid from the Early Cambrian. Nature 507, 496-500.
[12] Bambach R.K, Bush A.M. & Erwin D.H. (2007) Autecology and the filling of ecospace: key metazoan radiations. Palaeontology 50, 1-22.
[13] Erwin D.H. & Valentine J.W. (2013) The Cambrian Explosion: the construction of animal biodiversity. Roberts & Company Publishers. 406 pp.
The Encyclopedia of the Environment by the Association des Encyclopédies de l'Environnement et de l'Énergie (www.a3e.fr), contractually linked to the University of Grenoble Alpes and Grenoble INP, and sponsored by the French Academy of Sciences.
To cite this article: VANNIER Jean (January 5, 2025), The first complex ecosystems, Encyclopedia of the Environment, Accessed April 29, 2026 [online ISSN 2555-0950] url : https://www.encyclopedie-environnement.org/en/life/first-complex-ecosystems-2/.
The articles in the Encyclopedia of the Environment are made available under the terms of the Creative Commons BY-NC-SA license, which authorizes reproduction subject to: citing the source, not making commercial use of them, sharing identical initial conditions, reproducing at each reuse or distribution the mention of this Creative Commons BY-NC-SA license.




