Bats and the emergence of coronaviruses
PDF1. Most known coronaviruses come from bats

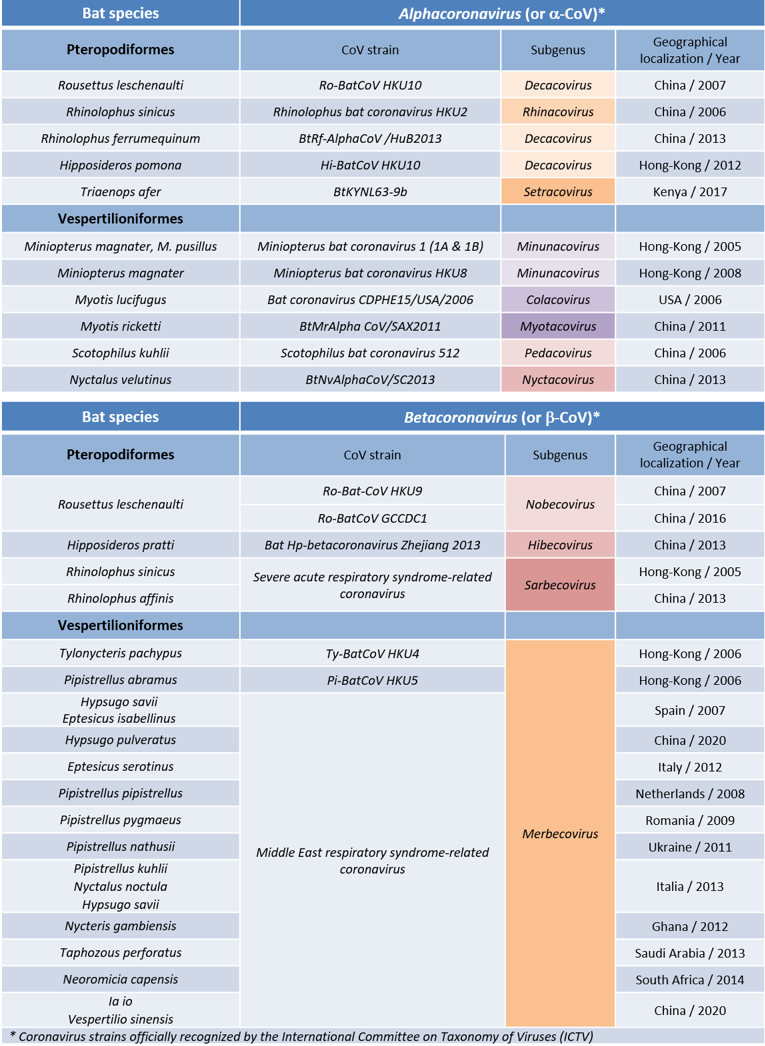
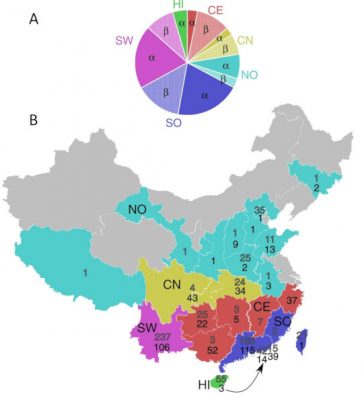
In the case of the SARS outbreak in 2002 – 2003, the masked palm civet and the raccoon dog (Nyctereutes procyonoides) were identified as the animals that transmitted the coronavirus (SARS-CoV-1) to humans. It was only much later that researchers discovered that this coronavirus originated from bat hosts belonging to species of the genus Rhinolophus, including Rhinolophus sinicus, R. ferrumequinum, R. affinis, R. macrotis, R. monoceros, R. pearsoni and R. pusillus (Figure 1). The geographical origin of these viruses has even been traced to a remote cave in Yunnan Province in south-western China.
Table. Bat coronaviruses, some of which are the cause of species responsible for recent epidemics.
Masked palm civets and raccoon dogs thus acted as intermediate hosts between bats and humans in transmission. Since the virus test was negative in civets living in the wild, it is likely that the human activities of raising civets, selling them at markets and eating them in restaurants were responsible for the contamination between wild animals and humans.
2. Where did the COVID-19 outbreak start?

Patient zero is yet to be found. Will we ever know him? If the disease is officially dated at the end of December 2019, analyses show that there will be patients in the Wuhan region as early as March 2019! In France, cases of COVID-19 could have already been present in mid-November 2019. We come to think that the first infection of a human being probably goes back several years. Studies have shown that viral transfers between wildlife and humans are much more frequent than previously thought and often go unnoticed. In the case of COVID-19, this is probably a single animal-to-human transmission event that was successful some time ago.
3. Origin of the SARS-CoV-2 coronavirus?
- Two genomes are 97% identical to each other and originate from bat viruses of the species Rhinolophus sinicus collected in 2015 and 2017 in Zhejiang province. They are 89% similar to SARS-CoV-2 [3].
- The third viral genome, BatCoV RaTG13, is much closer to SARS-CoV-2 with a sequence homology of 96.2%. This virus was isolated in 2013 from a bat of the same genus as before but belonging to a different R. affinis species recovered from an abandoned mine in Yunnan province [4].
Even if this percentage of homology seems high, we cannot confuse the two coronaviruses BatCoV RaTG13 and SARS-CoV-2, which certainly had a common ancestor whose separation is estimated to have occurred 40 to 70 years ago [5]. Recently, by reanalyzing the genome of a coronavirus called Ro-BatCoV HKU9 isolated in 2011 from a Leschenault’s rousette (Rousettus leschenaulti) from the Guangdong region, the researchers were challenged by the fact that by recombining the two viruses RaTG13 and HKU9, a virus very similar to SARS-CoV-2 was released [6]. It is not impossible that rousettes and horseshoe bats living in the same caves in southern China could have exchanged their coronaviruses, which in turn shared sequences of their genomes to produce a recombinant virus similar to SARS-CoV-2.
However, one mystery remains: how did SARS-CoV-2 end up in Wuhan in Hubei Province, which is far removed from the aforementioned provinces in southern China? This mystery is all the more complicated because the viral surveillance campaigns carried out in recent years among Hubei bats have not been able to isolate viruses related to SARS-CoV.
4. What were the circumstances of transmission of the pathogenic coronavirus to humans?
4.1. Direct contamination?
Contamination may have occurred directly during hunting activities involving bats. Hunters enter the caves and can contaminate themselves through contact with the faeces and aerosols generated by them. Contamination may also occur during the handling or transport of bat corpses, or during the cutting up of the bushmeat. However, while large fruit bats are a very popular dish, this does not seem to be the case for horseshoe bats who are too small to make a suitable meal. However, this cannot be ruled out.
The hypothesis of direct transmission of the virus to humans has been supported by seroprevalence studies that have shown the presence of bat coronavirus antibodies in the blood of a significant percentage of people living in rural areas of southern China. These results suggest that exchanges of coronavirus do take place between the inhabitants and bats grouped in colonies in caves near villages [7].
4.2. Intermediate hosts?

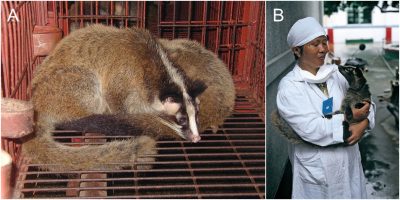
Feeding directly on bat corpses could be done by small carnivorous animals such as the Masked palm civet or the raccoon dog, already known as SARS-CoV-1 carriers, but also Asian badgers (Meles leucurus) and Chinese ferret-badgers (Melogale moschata). Haven’t we dismissed an obvious suspect, the masked palm civet [3] (Figure 5)? The Chinese are very fond of this animal and it has long been intensively farmed and had to be brought under control since the SARS epidemic in 2002. What about the measures taken for the current farms? We have little information.
In conclusion, there is a great deal of speculation about the origin of SARS-CoV-2 at this time. It should be remembered that it took 15 years to identify the bat virus that caused the SARS epidemic in 2002. It is highly likely that future discoveries will confirm the role of bats in the origin of COVID-19 [11]. However, it must be understood that these links between bats and viruses are not new; they may be very old. Just because this relationship has just emerged to our knowledge does not mean that it poses a greater threat now than it did before.
5. Messages to remember
- Most known coronaviruses are derived from bats in China.
- The coronaviruses causing SARS originate from bat hosts belonging to the genus Rhinolophus. Mutations have made these viruses contagious in humans after direct transmission or via intermediate hosts.
- For the SARS-CoV-2 responsible for the COVID-19 pandemic in humans, the chain of transmission is still the subject of much speculation.
- However, the newly emerging bat/virus linkages that we know of do not represent a greater threat now than before.
- In order to prevent new epidemics, it is imperative to address the risk factors that expose humans and wildlife.
Thanks to François Moutou and photographer Louis-Marie Préau (www.louismariepreau.com) for their contributions to this article.
Notes and References
Thumbnail photo. Greater horseshoe bat. [Source: © ML Préau, www.louismariepreau.com]
[1] Zhan SH, Deverman BE, Chan YA. (2020). SARS-CoV-2 is well adapted for humans. What does this mean for re-emergence ? bioRxiv preprint : https://doi.org/10.1101/2020.05.01.073262>.
[2] Latinne A, Hu B, Olival KJ, Zhu G, Zhang L, Li H, Chmura AA, Field HE, Zambrana-Torrelio C, Epstein JH, Li B, Zhang W, Wang LF, Shi ZL, Daszak P. (2020). Origin and cross-species transmission of bat coronaviruses in China. Nat Commun 11 (1), 4235. https://doi.org/10.1038/s41467-020-17687-3
[3] Li C, Yang Y, Ren L. (2020). Genetic evolution analysis of 2019 novel coronavirus and coronavirus from other species. Infect Genet Evol, 82, 104285. <doi: 10.1016/j.meegid.2020.104285>.
[4] Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, Chen HD, Chen J, Luo Y, Guo H, Jiang RD, Liu MQ, Chen Y, Shen XR, Wang X, Zheng XS, Zhao K, Chen QJ, Deng F, Liu LL, Yan B, Zhan FX, Wang YY, Xiao GF, Shi ZL. (2020). A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature, 579 (7798), 270-273. <doi: 10.1038/s41586-020-2012-7>.
[5] Boni MF, Lemey P, Jiang X, Lam TT-Y, Perry BW, Castoe TA, Rambaut A, Robertson DL. (2020). Evolutionary origins of the SARS-CoV-2 sarbecovirus lineage responsible for the COVID-19 pandemic. Nat Microbiol. < doi: 10.1038/s41564-020-0771-4>.
[6] Lau SK, Poon RW, Wong BH, Wang M, Huang Y, Xu H, Guo R, Li KS, Gao K, Chan KH, Zheng BJ, Woo PC, Yuen KY. (2010). Coexistence of different genotypes in the same bat and serological characterization of Rousettus bat coronavirus HKU9 belonging to a novel Betacoronavirus subgroup. J Virol, 84 (21),11385-11394. <Law: 10.1128/JVI.01121-10>.
[7] Li H, Mendelsohn E, Zong C, Zhang W, Hagan E, Wang N, Li S, Yan H, Huang H, Zhu G, Ross N, Chmura A, Terry P, Fielder M, Miller M, Shi Z, Daszak P. (2019). Human-animal interactions and bat coronavirus spillover potential among rural residents in Southern China. Biosaf Health, 1 (2), 84-90. <doi: 10.1016/j.bsheal.2019.10.004. Epub 2019 Nov 9>.
[8] Li X, Giorgi EE, Marichann MH, Foley B, Xiao C, Kong X-P, Chen Y, Korber B, Gao F. (2020). Emergence of SARS-CoV-2 through Recombination and Strong Purifying Selection. bioRxiv. preprint <doi: 10.1101/2020.03.20.000885>.
[9] Lee J, Hughes T, Lee M-H, Field H, Rovie-Ryan J J, Sitam, F T, Sipangkui S, Nathan S K.S.S, Ramirez Diana, Kumar S V, Lasimbang H, Epstein J H, Daszak P. (2020). No evidence of coronaviruses or other potentially zoonotic viruses in Sunda pangolins(Manis javanica) entering the wildlife trade via Malaysia. bioRxiv 2020.06.19.158717; doi: https://doi.org/10.1101/2020.06.19.158717
[10] Leroy EM, Ar Gouilh M, Brugère-Picoux J. (2020). The risk of SARS-CoV-2 transmission to pets and other wild and domestic animals strongly mandates a one-health strategy to control the COVID-19 pandemic. One Health, 100133. <doi: 10.1016/j.onehlt.2020.100133>.
[11] Ar Gouilh M, Puechmaille SJ, Diancourt L, Vandenbogaert M, Serra-Cobo J, Roïg ML, Brown P, Moutou F, Caro V, Vabret A, Manuguerra JC, EPICOREM consortium. (2018). SARS-CoV related Betacoronavirus and diverse Alphacoronavirus members found in western old-world. Virology, 517, 88-97. <doi: 10.1016/j.virol.2018.01.014>.