Why and how to treat urban wastewater?
PDF
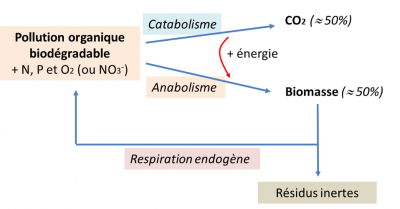
Faced with the increase in the world’s population combined with increasing urbanization, access to drinking water and sanitation is still a vital issue for many cities, especially in developing countries. Sanitation refers to all the techniques used to collect, transport and treat wastewater before it is discharged into the natural environment. It can be conceived on the scale of an urban area (collective sanitation) or of dwellings not connected to a collective sewer network (autonomous sanitation). The wastewater treatment plants of the future tend to become real wastewater treatment plants in order to produce green energy, fertilizing materials and precious metals and to reuse treated wastewater.
1. A brief history of urban sanitation

Following successive cholera epidemics that swept the world during the 19th century, the hygienist movement advocated in the 1850s the construction of underground sewer systems (Figure 2) to discharge domestic sewage, stormwater and street water directly into rivers or the sea. The length of the City of Paris’ sewerage network has thus increased from 150 km in 1853 to nearly 900 km in 1890 (currently about 2500 km). In 1894, a law forced Parisian buildings to discharge their waste and rainwater as well as their black water [1] into the newly created (so-called combined) sewer system [2]. The concept of the sewerage system thus appeared.

Wastewater disposal shifted the problem of nuisance out of the cities and generated increasingly unacceptable pollution of surface water. The first purification techniques appeared in the 1860s with the spreading of raw wastewater on sandy soils in order to use the purifying power of the soil while increasing agricultural and market gardening production.
The continuous increase in urbanization and the volume of wastewater collected has led to a concomitant increase in the surface area of land application fields (up to 5000 ha for Paris around 1900). Between 1870 and 1900, spreading capacity was gradually increased by draining fields and removing solids before spreading by settling, chemical treatment or anaerobic fermentation (i.e. in the absence of oxygen).
In the 1880s, artificial filters with high porosity and made of materials (coke, bottom ash, pozzolana, etc.) appeared, encouraging the intensive development of a biofilm [3] purifier. These are the bacterial beds. The first of them was built in 1893 in Salford (England).
In 1914, English researchers Ardern and Lockett discovered that pollution control was much faster when the wastewater to be treated was brought into contact with an already formed purifying biomass [4]. They thus file the first patent on the purification process, which will be called the activated sludge process. This process, which does not use a filter, is based on an intensive culture of a purifying biomass suspended in water (activated sludge). It was first implemented industrially in England in 1914 with a single reactor (with a sequenced feed) and then in 1916 with a biological reactor continuously fed and coupled to a settling tank (or clarifier).
These processes have been regularly improved up to the present day thanks to the progress made in electromechanical equipment and the new scientific knowledge acquired since the 1970s in understanding and optimising the reactions to eliminate nitrogen and phosphorus pollution. While many bacterial beds and activated sludge plants were built in the 1920s and 1960s, it was not until the 1970s that the construction of wastewater treatment plants in developed countries really took off due to an increasing collective awareness of the need to protect the environment, supported by the publication of increasingly restrictive regulatory texts (read Water law in France).
2. Why treat urban wastewater?
2.1. Composition of urban wastewater
Urban wastewater contains a large number of organic and inorganic compounds found in black water (loaded with urine and faeces), dirty water from food preparation, laundry and bathrooms and in runoff water. From an analytical and regulatory point of view, the characterization of raw and treated wastewater uses global parameters (expressed in mg/l) that include a set of pollutants:
- The content of suspended matter (SM), which represents the particulate pollution retained by filtration through 1.2 µm porosity filters. They are composed of approximately 25% mineral matter and 75% organic matter known as volatile matter in
- Chemical oxygen demand (COD) is the amount of oxygen required to fully oxidize dissolved and particulate organic pollution, which is biodegradable and non-biodegradable to CO. This total oxidation is carried out chemically by using a very strong oxidant (potassium dichromate), in a very acidic environment and at about 150°C for 2 hours [5]. For raw domestic wastewater, about 50% of the COD is in dissolved form and 50% in particulate form.
- Biological oxygen demand over 5 days (BOD5) is the amount of oxygen consumed by bacteria after 5 days of reaction to degrade biodegradable organic matter. The COD/BOD5 ratio of raw urban wastewater (2 to 2.5) indicates that organic pollution can be easily removed biologically in a wastewater treatment plant.
- Nitrogen Kjeldahl [6] (NK) takes into account the total content of organic nitrogen (included in urea, amino acids, proteins…) and ammoniacal nitrogen (N-NH3).
- Nitrogen global (NGL) is the total content of organic nitrogen, ammonia nitrogen, nitrous nitrogen (N-NO2-) and nitric nitrogen (N-NO3-). The latter two forms of nitrogen are not present in raw urban wastewater.
- Total phosphorus (Pt) includes organic and inorganic phosphorus.
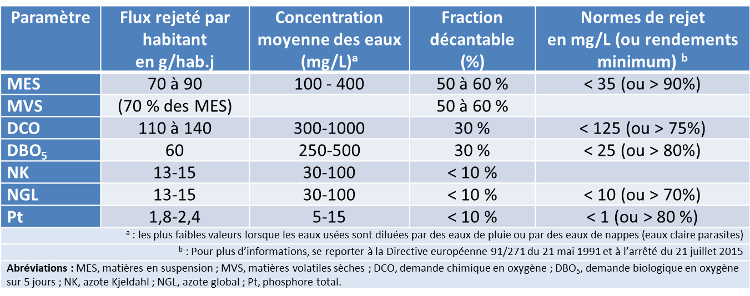
Table 1 gives the average characteristics of urban wastewater at the entrance to the treatment plant as well as the minimum quality of treated wastewater required by the regulations (maximum concentrations or minimum disposal efficiencies).
Table 1. Average composition of raw urban wastewater and examples of discharge standards for a large wastewater treatment plant (for more than 100,000 inhabitants).
Urban wastewater also contains many inorganic and organic compounds at much lower concentrations (from ng/l to a few µg/l). The main families of micropollutants are cosmetics, pesticides and pesticide residues, solvents, natural and synthetic hormones, drug residues, metals, etc. This micro-pollution is the subject of regulatory action campaigns for the release of substances dangerous to the environment. Currently, particular attention is being paid to the concentrations of pesticide residues, drugs and endocrine disrupters in wastewater entering and leaving wastewater treatment plants.
Urban wastewater also contains a high concentration of microorganisms of faecal origin and in particular pathogenic microorganisms, the number and type of which depend on the health status of the population.
2.2 Impacts of releases on aquatic environments
The discharge of untreated urban wastewater into surface water causes visual pollution (floating matter), reduced water transparency and siltation of lakes and rivers. The discharge of biodegradable substances promotes biological activity in watercourses, which leads to a decrease in dissolved oxygen concentration and even asphyxiation of the watercourses. Nitrogen and phosphorus discharges promote eutrophication phenomena (read Phosphorus and Eutrophication and Nitrates in the Environment).
The release of micropollutants can have toxic effects on the fauna and flora of aquatic environments. These effects include the bioaccumulation of persistent molecules in the food chain, chronic toxicity at very low doses and changes in the functioning of the endocrine system, which can result, for example, in the feminization of male fish. Microbiological water pollution can make water quality unfit for certain uses.
2.3. Obligation to treat waste water
Microorganisms naturally present in surface water can degrade polluting substances brought by wastewater discharges, but the self-purifying capacities of rivers are generally very much insufficient. Wastewater must therefore be treated in treatment plants before being discharged into the natural environment. For the various global pollution parameters, the maximum concentration values not to be exceeded in treated wastewater or the minimum purification efficiencies to be achieved are set by regulatory texts, see Table 1 (see also Water law in France).
3. How is wastewater treated?
The biological elimination of organic, nitrogen and phosphorus pollution requires, for each type of reaction, specific conditions for implementation (presence or absence of dissolved oxygen, residence time of biomass in reactors, etc.). The purification is carried out by cultures of purifying biomass suspended in water or fixed on a support.
3.1. Biological transformation of pollution
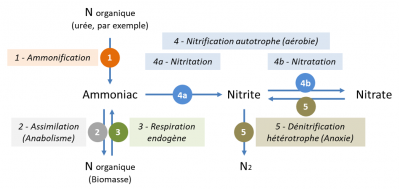
- Ammonification (reaction 1) converts organic nitrogen (mainly contained in urea for wastewater) into ammonia nitrogen. This reaction is rapid; it is ensured by many types of microorganisms:
Urea[CO(NH2)] → Ammonia[NH3] + Carbon dioxide[CO2]
- The assimilation (reaction 2) of ammoniacal nitrogen by bacteria to form new organo nitrogen biomolecules used in the composition of new bacteria.
- Biological nitrification (reactions 4a and 4b) converts ammoniacal nitrogen (ammonium, NH4+) into nitrous nitrogen (nitrite, NO2-) by nitriting bacteria and then into nitric nitrogen (nitrate, NO3-) by nitrating bacteria:
Ammonium[NH4+] → Nitrite[NO2-] → Nitrate[NO3-]
These reactions occur only in the presence of oxygen. They are carried out by so-called autotrophic bacteria because they use mineral carbon (CO2 or HCO3-) as a carbon source for the synthesis of new bacteria.
- Biological denitrification (reaction 5) reduces nitrate ions (NO3-) to nitrous oxide (N2). In wastewater treatment plants, denitrification is only possible in the absence of oxygen. It is carried out by heterotrophic bacteria and requires the presence of organic matter. As illustrated by the written reaction for denitrification in the presence of methanol (a small, highly biodegradable organic molecule), denitrification is accompanied by the elimination of organic pollution (oxidation to CO2):
Nitrate[NO3-] + Methanol[CH3OH] → Nitrogen[N2] + Carbon Dioxide[CO2] + Water[H2O]
As with ammonia nitrogen, biomass growth is accompanied by partial removal of phosphorus by assimilation (incorporation of phosphorus into new biomolecules).
Further biological phosphorus removal is only possible if the biomass undergoes an alternation of anaerobic and aerobic phases allowing the development of so-called dephosphating bacteria that have the property of overaccumulating phosphorus in their cells. Phosphorus can thus represent 10 to 12% of the dry weight of dephosphating bacteria compared to 1 to 2% for non-dephosphating bacteria.
In wastewater treatment plants, biological phosphorus removal only removes about 40 to 60% of the phosphorus. In order to meet the discharge standards (see Table 1), it is supplemented by physico-chemical dephosphatation, which consists of the precipitation of phosphorus in the form of ferric phosphate by the addition of an iron salt (usually ferric chloride, FeCl3).
3.2. Intensive processes for large wastewater treatment plants

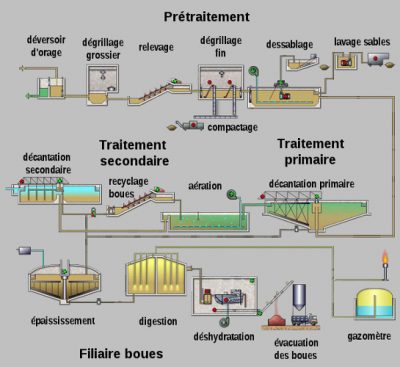
- pre-treatment steps to remove large debris (screening and sieving), sand (desanding) and grease (degreasing);
- activated sludge reactor(s)
- a settling tank (called a clarifier)
- possibly tertiary treatment (e. g. disinfection by UV irradiation) before release into the natural environment.
An activated sludge wastewater treatment plant generally consists of two or three biological reactors in series, the layout and operating conditions of which have been chosen in order to optimise the removal rates of organic, nitrogen and phosphorus pollution. The formation of bacterial flocs in activated sludge tanks separates treated wastewater that overflows and sludge that settles at the bottom of the structure by settling in the clarifier (Figure 8). Some of the sludge is recirculated in the biological reactor(s) and the excess is sent to the sludge treatment and recovery process. Invented in 1914, this type of wastewater treatment plant treats about 90% of the urban waste collected in France.

Biofiltration purification plants. This process, developed in the 1980s, eliminates organic and nitrogen pollution by a fixed biomass that develops in a submerged filter. This medium (grain size: 4 to 6 mm) acts both as a support for the development of the biofilm and as a filter medium. At the outlet, the treated water can be discharged directly into the receiving environment (no clarifier). In order not to clog the biofilters too quickly, a thorough removal of suspended solids is carried out before biofiltration. A daily washing of the biofilters makes it possible to recover the suspended matter retained as well as the biomass produced during filtration.
3.3. Extensive processes for small wastewater treatment plants

Natural lagooning consists in circulating the wastewater to be treated for several tens of days in 3 watertight basins (lagoons) in series of shallow depths (1 to 1.4 m) (Figure 9).
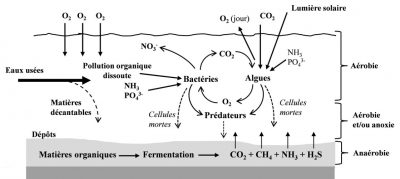
- Upper water layer: under aerobic conditions, bacteria oxidize biodegradable (especially in the 1st basin) and nitrogenous (nitrification 2nd and 3rd basin) organic pollution. Dissolved oxygen is provided by the exchanges between water and the atmosphere by microalgae (photosynthesis). During their development, microalgae assimilate part of the nitrogen and phosphorus and phosphorus provided by wastewater.
- Basically: the absence of dissolved oxygen at the bottom of the tanks (no photosynthesis) allows partial denitrification of nitric nitrogen and possibly leads to anaerobic fermentation reactions (production of hydrogen sulphide).
The purifying performance of lagooning varies according to the seasons according to variations in sunshine and water temperature. The basins are cleaned every 10 to 12 years due to sediment accumulation.

4. The wastewater treatment plants of the future: real plants to be recycled
The main objective of wastewater treatment plants is to reduce the flow of pollution discharged into the natural environment. They can also become real plants to produce green energy, raw materials or to reuse treated water [11], [12]. These new actions are part of the sustainable development, circular economy, renewable energy production and global warming initiatives developed by cities and local authorities.
4.1. Green energy production
Each inhabitant connected to a wastewater treatment plant produces about 20 to 25 kg/year (in dry weight) of wastewater sludge. This sludge, which contains about 65% organic matter, nitrogen and phosphorus, has long been used in agriculture (spreading, composting). They are increasingly used in large wastewater treatment plants for the production of renewable energy. Sludge incineration (alone, with household waste or in cement plants) reduces the consumption of fossil fuels.
The anaerobic digestion of sewage sludge produces about 6 m3 of biogas/H.E. per year (containing 65% methane and 30% CO2). Biogas can be upgraded to produce heat and/or electricity or injected into the city gas grid after purification. The methanisation of sewage sludge is a proven process and the optimisation of the recovery of the biogas produced is currently being sought in new installations.
If the waterfall and water flow permit, turbines can be installed in pipelines upstream or downstream of a sewage treatment plant to convert hydraulic energy into electricity. Switzerland can thus produce at least 9.3 GWh/year of hydroelectric power from wastewater in a cost-effective manner and 3.5 GWh/year are already being used [13].
Wastewater heat [14] is a renewable source of energy [15]. Heat can be recovered from the sewerage network or from wastewater treatment plants using heat exchangers to supply heat pumps that produce heat and cooling on a local scale. Wastewater heat recovery is currently experiencing a very significant expansion for domestic hot water production, heating and air conditioning in public buildings, swimming pools and buildings. Pilot studies are currently being conducted to grow microalgae crops using nitrogen and phosphorus from treated wastewater as a nutrient source to produce biofuels (read Biofuels: Is the future in microalgae?).
Biopiles are reactors that use bacteria to directly convert the energy released during the oxidation of biodegradable compounds into electricity. Current research is attempting to develop biopiles that are technically and economically viable.
4.2. Production of raw materials
Phosphorus contained in wastewater can be precipitated (in the form of calcium phosphate or struvite granules [16]) and reused as a fertilizer in agriculture. The results of one of the first industrial projects (Aarhus wastewater treatment plant in Denmark, 85 000 p.e.) indicate that 60% of the 700 to 750 g of phosphorus released annually by each inhabitant can be recycled, thus limiting the import of phosphates extracted from ores.
Wastewater contains precious and rare metals such as copper, silver, gold, platinum, palladium, vanadium,…. According to researchers at Arizona State University (USA), there is the equivalent of $13 million worth of metals in sewage sludge produced each year by a city of one million people, including $ 2.6 million in gold and silver. These metals are found in sewage sludge [17]. In Japan, a wastewater treatment plant is already recovering gold from the incineration ashes of sewage sludge [18].
Recent small-scale studies have shown that it is possible to produce a biodegradable bioplastic (a polyhydroxyalkanoate: PHA) using bacteria that transform organic matter in raw wastewater or sewage sludge. A wastewater treatment plant receiving wastewater from 1 million inhabitants can potentially produce 18,000 tonnes of PHA/year.
4.3. Reuse of treated wastewater
Many parts of the world suffer from a temporary or even chronic water deficit and treated wastewater can be reused to compensate for the lack of water resources. They can be used for watering green spaces and golf courses, irrigation of agricultural areas, industrial needs or for the production of drinking water [19] (direct reuse in a drinking water production plant or indirectly by recharging water reservoirs or water bodies by infiltration). Depending on the destination of the water, wastewater leaving a treatment plant must undergo appropriate after-treatment, ranging from simple disinfection to a succession of much more complex treatments.
5. Messages to remember
- Wastewater treatment was first introduced for health reasons in large urban centres, and then began to expand in the 1970s to meet stricter standards for the preservation of natural environments.
- Urban wastewater is treated by biological processes, supplemented by physico-chemical dephosphatation. Industrial polluted water is treated separately in specific installations.
- For urban areas with more than 2000 to 4000 inhabitants, intensive wastewater treatment plants mainly use the activated sludge process (90% of urban water collected in France).
- For small communities, extensive wastewater treatment plants use natural lagooning or reed filters.
- Ongoing developments aim to recover sewage sludge, by recovering energy in the form of biogas or heat, producing fertilizers or bio-plastics, and sometimes by recovering extracted metals.
References and notes
Cover image. Antwerp-Zuid wastewater treatment plant. [Source: By Annabel[CC BY-SA 3.0 (https://creativecommons.org/licenses/by-sa/3.0)], from Wikimedia Commons.]
[1] Heavily loaded water from toilets.
[2] Tabuchi J.P. (2008) L’assainissement de l’agglomération parisienne, Nanterre, Agence de l’eau Seine-Normandie, 35 p. http://www.reseauprojection.org/ateliers/niamey_2009/Documents_annexes/session%203%20-%20Histoire%20assainissement%20agglo.%20parisienne%20JPT.pdf
[3] A biofilm is a community of microorganisms (bacteria, fungi, algae or protozoa), adhering to each other and to a surface, and marked by the secretion of an adhesive and protective matrix. It is usually formed in water or in an aqueous medium.
[4] The purifying biomasses that develop in activated sludge are composed of a mixture of living or dead microorganisms (Bacteria & Actinomycetes), organic and/or mineral debris, colloids and a microfauna composed of small animals, specific to the site.
[5] For example, plant debris (particulate pollution) is oxidized in an acidic medium by potassium dichromate to CO2 and therefore consumes oxygen (1 mole of dichromate consumed = 1.5 mole of O2).
[6] The Kjeldahl method is a technique for determining the nitrogen level in a sample developed in 1883 by the Danish Kjeldahl. Largely modified later, it is known as the determination of nitrogen following Kjeldahl, Wilforth and Gunning.
[7] Molecules excreted by microorganisms and which allow their aggregation, for example in biofilms.
[8] Wastewater contains a large proportion of organic matter composed of colloids that cannot aggregate because of their electronegative charge. To destabilize this suspension and recover the compounds, it is necessary to promote the agglomeration of colloids by reducing their electrostatic repulsion forces. This is the coagulation phase. The micro-flocs aggregate with each other to form larger flakes, until they become settleable by gravity. The floc is thus formed.
[9] PE = Inhabitant Equivalent. A unit of measurement used to evaluate the capacity of a wastewater treatment plant based on a quantity of pollution emitted per person per day equal to 60 g BOD5 per day. (Example: a treatment plant of 1000 p.e. receives 60 kg BOD5/day).
[10] The reduction of bacteria corresponds to a reduction in the number of bacteria in the environment after treatment.
[11] Found E. (2010). The wastewater treatment plant of the future. Le cahier des chroniques scientifiques de Véolia, n°17, 13 pages.
[12] ARMC, Rhone Mediterranean Corsica Water Agency (2016). The wastewater treatment plant of the future. She’s resourceful! (5-page brochure).
[13] Bousquet C., Samora I., Manso P., Schleiss A., Luca Rossi L., Heller P. (2015). Wastewater turbining. What potential for Switzerland? Aqua and Gas, 15,54-61.
[14] The temperature of the wastewater is between 12°C and 20°C depending on the time of day and the seasons.
[15] De Batz S., Van den Bossche H. (2007). The production of renewable energy by purification systems, some examples. Techniques Sciences and Methods, 12, 67-83.
[17] Westerhoff P., Lee S., Yang Y., Gordon G.W., Hristovski K., Halden R.U., Herckes P. (2015). Characterization, recovery opportunities, and evaluation of metals in municipal sludges from U.S. wastewater treatment plants nationwide. Environmental Science & Technology, 49(16), 9479-9488.
[18] A wastewater treatment facility in Suwa, near Nagano (central Japan), yielded 1,890 grams of gold per tonne of ash from incinerated sludge. The high percentage of gold found is probably due to the large number of precision equipment manufacturers in the area. This gold content is much higher than that of gold mines. For example, the Japanese Hishikari mine (Sumitomo Metal Mining Co Ltd) produces 20 to 40 grams of precious metal per tonne of ore.
[19] J. Haarhoff, B. Van der Merwe (1996) Twenty-five years of wastewater reclamation in windhoek, Namibia, Water Science and Technology 33 (10-11) 25-35;
The Encyclopedia of the Environment by the Association des Encyclopédies de l'Environnement et de l'Énergie (www.a3e.fr), contractually linked to the University of Grenoble Alpes and Grenoble INP, and sponsored by the French Academy of Sciences.
To cite this article: DE LAAT Joseph (January 5, 2025), Why and how to treat urban wastewater?, Encyclopedia of the Environment, Accessed May 27, 2026 [online ISSN 2555-0950] url : https://www.encyclopedie-environnement.org/en/water/why-how-treat-urban-wastewater-2/.
The articles in the Encyclopedia of the Environment are made available under the terms of the Creative Commons BY-NC-SA license, which authorizes reproduction subject to: citing the source, not making commercial use of them, sharing identical initial conditions, reproducing at each reuse or distribution the mention of this Creative Commons BY-NC-SA license.