Bats and viruses or how to live together in harmony
PDF
Bats have long been ignored or even despised, and in recent years they have been the subject of increasing public and research attention. Endowed with exceptional abilities such as flight and echolocation, bats intrigue the former. Their unexpected longevity given their size and, above all, a unique immune system that enables them to tolerate many viruses that are poorly tolerated by terrestrial mammals, make them excellent study models for the latter. However, this improved image is tarnished by the knowledge of their links with pathogenic viruses. Indeed, bats seem to play a crucial role in the emerging virus epidemics that have been shaking the world population with increasing frequency in recent years. The current COVID-19 pandemic is no exception, and has once again brought bats to the forefront. This raises many questions: How are bats preferred hosts for pathogenic viruses? What role did they play in the origin of the COVID-19 pandemic? Are they indeed responsible for the emergence of the new viral diseases that are increasingly affecting humans and domestic animals?
- 1. Bats in the spotlight
- 2. Bats and viruses?
- 3. Why are bats preferred hosts for pathogenic viruses?
- 3.1. Viruses and bats have been living together for a long time
- 3.2. A long period of co-evolution between viruses and bats
- 3.3. Flying, essential to the links between bats and viruses
- 3.4. Breathing and oxidative stress
- 3.5. Exceptional longevity conducive to virus persistence
- 3.6. Lethargy and torpor
- 3.7. Importance of living together
- 3.8. Unique immune abilities
- 4. Are bats responsible for viral outbreaks?
- 5. Messages to remember
1. Bats in the spotlight
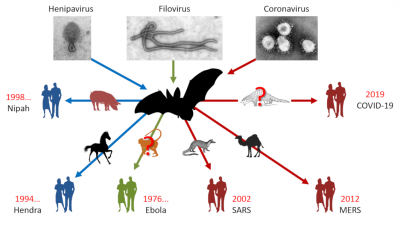
As with SARS-CoV-1, bats appear to be involved in the viral transmission chain. Indeed, preliminary results have shown that SARS-CoV-2 is thought to result from the recombination of a bat coronavirus [1] with a Malayan pangolin (Manis javanica) coronavirus [2]. The latter, like the masked palmed civet (Paguma larvata) in the 2002-2003 epidemic, is believed to have acted as an intermediate host between bats and humans to give rise to SARS-CoV-2, which is highly contagious in humans (See Focus: Bats and coronavirus emergence).
This is not the first time that bats have been in the spotlight. We have to go back to 1911 for the first mention of a virus isolated from bats. At the time, rabies was the subject of interest and it was discovered that it was transmitted by the common vampire bats of Brazil. After a long period of oblivion, it was rediscovered during the years 1990-2000 in the framework of health surveillance programmes that bats are the natural hosts of many emerging viruses such as henipaviruses (Hendra and Nipah), filoviruses (Ebola) and coronaviruses (SARS) [3] (Figure 1). Unexpectedly, they are found to be unaffected by most of these pathogens. It has even been demonstrated, particularly for the Hendra and Nipah viruses, that they can withstand viral loads that are normally lethal in other mammals [4].
2. Bats and viruses?
2.1. Bats account for a quarter of all mammalian species
A recent revision of the order of Chiroptera based on molecular data has recast it into two new suborders that are more phylogenetically coherent (See: Focus Bats and Coronavirus Emergence) :

- Yinpterochiroptera (Pteropodiformes), which includes tropical fruit bats or flying foxes (Figure 3);
- The Yangochiroptera (Vespertilioniformes) which includes all microchiroptera except five families.
2.2. A very wide variety of ecological niches

2.3 What are bat viromes?
Thanks to these samples, a large number of viral sequences were identified, some of which were found to be similar to those of many viruses known to be pathogenic to humans [6]. Nevertheless, beware not to conclude, as has sometimes been done rather hastily, that bats are responsible for all the viral diseases that affect humans! Although these methods have brought significant advances in our knowledge of the links between viruses and bats, we must nevertheless remain cautious in interpreting the considerable mass of data obtained, given the weakness of the means of analysis.
However, it is surprising to note that all these advances have confirmed simple observations made in 1974, which already revealed the propensity of bats to host a wide variety of RNA and DNA viruses [7].
2.4. Are bats “virus strain”?
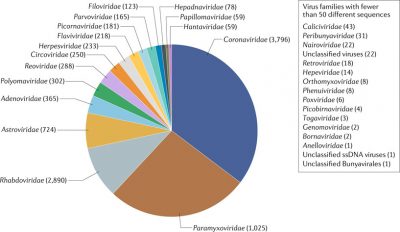
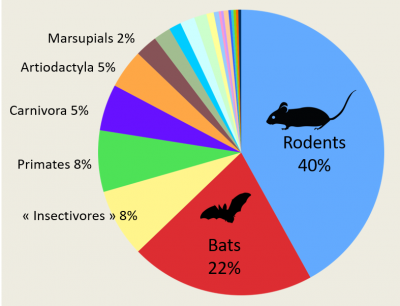
With regard to coronaviruses in particular, another large comparative study [10] was conducted in more than 19,000 animals belonging to bats, rodents and non-human primates. It showed that 98% of the coronaviruses found came from bats. A massive and diverse presence since, out of one hundred types of coronavirus identified, 91 came from Chiroptera. The proportion of individuals positive to a coronavirus was 8.6% in bats and 0.2% in other animals. Extrapolating these results to all species of bats, they would be carriers of no less than 3,200 coronaviruses throughout the world. This number was recently re-evaluated at 3,796 (Figure 6) [11]. Bats therefore seem to have special abilities.
Nevertheless, all this should be put into perspective because sampling bias exist in these studies. The order of Chiroptera, with its 1,428 species, occupies the second place among Mammals, and bats are inevitably at the forefront of the scene. Recently, researchers have shown that by going beyond mammals and including birds, the risk of viral zoonoses remains homogeneous regardless of whether the host species belong to the avian or mammalian orders, and thus the number of zoonoses per taxonomic order increases with species abundance [12]. Thus, for bats and rodents, the high number of zoonotic viruses is simply related to the specific richness of the two orders they represent.
In fact, explanations of the special role that bats play in viral emergences are more likely to be sought in terms of their particular ecoethology, their unusual physiological and immune characteristics, and also their ability to exchange RNA viruses more intensely than in other animal species. Finally, we must add the way in which humans, through their action on ecosystems, create favourable conditions for bringing all these elements into synergy.
3. Why are bats preferred hosts for pathogenic viruses?
3.1. Viruses and bats have been living together for a long time

3.2. A long period of co-evolution between viruses and bats
Viruses have thus had a very long time to adapt to their Chiropteran hosts and co-evolve intimately. A slow selection of the viruses’ capacities to infect their host took place, leading to increasingly close viral adaptation to certain molecules present on the surface of bat cells. Since these molecules act as receptors, they will play a crucial role in the penetration of the virus into the host cells. Moreover, being involved in a number of fundamental physiological processes, these receptor molecules have remained highly conserved in the animal world. This explains the ease with which the viruses play on species barriers and circulate rapidly among mammalian hosts.
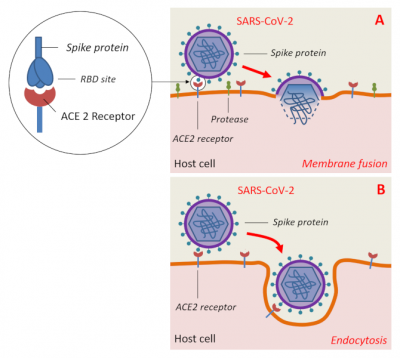
However, the key (S protein) must still have mutagenically acquired a specific amino acid sequence in its receptor binding domain (RBD) to the human ACE2 receptor as well as a specific site of cleavage by host enzymes so that SARS-CoV-2 may infect human cells [14].
These mutations were presumably acquired by the bat coronavirus during passage through intermediate hosts to result in SARS-CoV-2. In addition to humans, the latter is capable of infecting other mammals such as primates (Macaque monkeys), certain species of deer and cetaceans, domestic cats, tigers, golden hamsters, ferrets, mink and domestic dogs. Apart from Mammals, other classes of vertebrates do not seem to be affected [15].
3.3. Flying, essential to the links between bats and viruses

More unexpectedly, it was realized that flight, by greatly increasing the metabolism (15 to 16 times compared with the 2 times of most birds) and the bat’s body temperature (>38°C), induced a response similar to that of a feverish state. However, fever is a natural defence mechanism used by endothermic animals to inhibit the growth of pathogens and stimulate their immunity. Thus, by inducing a more or less permanent feverish state, flight would contribute to making bats more resistant to viral attacks [16].
3.4. Breathing and oxidative stress
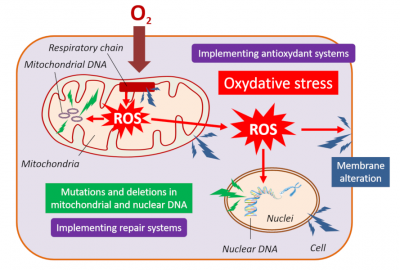
3.5. Exceptional longevity conducive to virus persistence
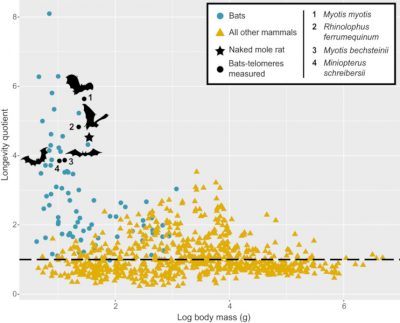
We saw earlier that the bat manages to moderate oxidative stress, which generally leads to chronic inflammation and accelerated aging. Similarly, the progressive shortening of telomeres (chromosome ends), which normally causes senescence and the appearance of cancer, has not been observed in bats [20]. Bats thus show exceptional longevity (Figure 11) with few signs of senescence and a negligible rate of cancer. Out of 19 mammalian species that live proportionally longer than humans, taking their body size into account, 18 are bats, with a record held by a Brandt’s bat (Myotis brandtii), which, at a weight of 7 g, has exceeded the age of 41 years. This longevity is conducive to the persistence of viruses in individuals and to long-term viral exchanges between generations of juveniles and adults.
3.6. Lethargy and torpor

Lethargy is a strategy used by bats in temperate countries to survive the insect shortage that occurs during cold seasons while other animals such as birds choose to migrate to warmer climates.

3.7. Importance of living together
Viral persistence can also be expressed on an individual level, as well as on a collective level. Chiropterans have a strong propensity to group together in colonies of hundreds or even thousands of individuals of different ages and sometimes of different species. The largest known concentration of mammals is found in the Braken Cave in Texas, which concentrates more than 20 millions of Brazilian free-tailed bats (Tadarida brasiliensis mexicana) (Figure 14). It takes at least 3 hours for the entire colony to emerge from the cave!

In these groupings, it has been well shown that the nature and size of colonies play an important role in viral maintenance and the appearance of new variants. It is thought that viral exchanges between adults and juveniles promote chronicity of infection, whereas between individuals of different species, viral diversity increases. Researchers are now wondering whether this habit of living in sympatry may be the determining factor that has led bats to tolerate viruses while developing unique immune capacities, aspects of which have only recently begun to be identified.
3.8. Unique immune abilities
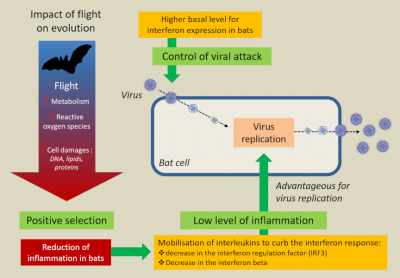
In fact, bats seem capable of restricting their immune response to pathogens with which they share a long evolutionary history in order to limit the immunopathological consequences of an infection [22]. A kind of non-aggression pact seems to have been established between them and viruses.
Recent analysis of six bat genomes [13] has shown a wide variety of endogenous viruses, which tends to confirm this state of tolerance towards viruses recognised as elements of the bat self. It also revealed the concentration of the inflammatory response on a limited number of genes and the existence of new genes involved in tolerance to viral infections. This study aims to better understand how bats tolerate coronavirus infections.
The recognition of the role of bats in viral epidemics presents the risk of bats being responsible for them and thus of considering their eradication as a solution to the risk of infection. This reaction, which is unfortunately intuitive, would prove to be totally inappropriate and even prejudicial to human health. It has already been tested in Uganda where, as part of campaigns to prevent Marburg virus infections, destruction of fruit bats has been carried out in some mines. This has resulted in re-invasion of these sites by susceptible bats and multiple reintroductions of the virus into new connected populations.
Reacting like this means forgetting that Chiropterans are key species in the functioning of ecosystems. In tropical environments, they play a significant role in the pollination of plants and the long-distance dissemination of seeds. Humans benefit by consuming them and using their guano as fertilizer. As for insectivorous bats, they play a major role in regulating insect populations and thus contribute to a reduction in the use of pesticides. Very sensitive to changes in their environment, bats are excellent indicators of the health of our environment.
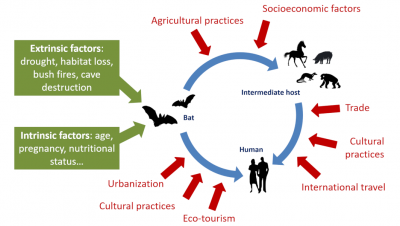
In a more comprehensive way, it will be necessary to act on the risk factors that expose humans to wildlife and bats. The COVID-19 pandemic is there to remind us of this. It made it clear that controlling these risks requires close collaboration between the fields of human health, animal health and ecosystem health. In other words: doctors, pharmacists, veterinarians and ecologists, as well as economists and lawyers, must work together to promote an ecology of health, an essential approach to prevent future epidemics and prevent them from turning into health crises (See Viral pandemics of the modern era).
5. Messages to remember
- Bats are natural hosts for many emerging viruses, but are unaffected by most of them.
- Bats are the preferred hosts of viruses because of their diversity of species, 2nd order in Mammals, diets, and ecological niches.
- Research is being conducted on the virome (set of viral genomes) of bats, which shows that they are not directly responsible for human viral diseases.
- Their appearance dates back more than 65 million years, which has given them time to co-evolve closely with viruses.
- The success of Chiropterans is due to their unique adaptations: flight and echolocation abilities, exceptional longevity, ability to enter states of lethargy or torpor, gregarious instinct and unique immune system that have contributed to their wide tolerance to infections
- Chiropterans are key species in the functioning of ecosystems and excellent indicators of the health of our environment.
- The causes of epidemics are rather to be found in the disruption of natural ecosystems due to uncontrolled human activities.
- To prevent new epidemics, it is imperative to act on the risk factors that expose humans and wildlife.
Thanks to François Moutou for his proofreading of the article and his exotic photographs, and to Marc Pihet and Louis-Marie Préau (www.louismariepreau.com) for their photographic contributions illustrating this article.
Notes and References
Cover image. Greater mouse-eared bat (Myotis myotis) in flight. [Source: © Louis-Marie Préau, www.louismariepreau.com]
[1] Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, Chen HD, Chen J, Luo Y, Guo H, Jiang RD, Liu MQ, Chen Y, Shen XR, Wang X, Zheng XS, Zhao K, Chen QJ, Deng F, Liu LL, Yan B, Zhan FX, Wang YY, Xiao GF & Shi ZL. (2020). A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature, 579 (7798), 270-273. <doi: 10.1038/s41586-020-2012-7>.
[2] Xiao K, Zhai J, Feng Y, Zhou N, Zhang X, Zou J-J, Li N, Guo Y, Li X, Shen X, Zhang Z, Shu F, Huang W, Li Y, Zhang Z, Chen R-A, Wu Y-J, Peng S-M, Huang M, Xie W-J, Cai Q-H, Hou F-H, Chen W, Xiao L & Shen Y. (2020). Isolation of SARS-CoV-2-related coronavirus from Malayan pangolins. Nature, 583 (7815), 286-289. <doi: 10.1038/s41586-020-2313-x>.
[3] Calisher CH, Childs JE, Field HE, Holmes KV & Schountz T. (2006). Bats: important reservoir hosts of emerging viruses. Clin. Microbiol. Rev., 19 (3), 531-545. <doi:10.1128/CMR.00017-06>.
[4] Middleton DJ, Morrissy CJ, van der Heide BM, Russell GM, Braun MA, Westbury HA, Halpin K & Daniels PW. (2007). Experimental Nipah virus infection in pteropid bats (Pteropus poliocephalus). J. Comp. Pathol., 136 (4), 266-272. <doi: 10.1016/j.jcpa.2007.03.002>.
[5] Hu D, Zhu C, Wang Y, Ai L, Yang L, Ye F, Ding C, Chen J, He B, Zhu J, Qian H, Xu W, Feng Y, Tan W & Wang C. (2017). Virome analysis for identification of novel mammalian viruses in bats from Southeast China. Sci. Rep. 7 (1), 10917. <doi: 10.1038/s41598-017-11384-w>.
[6] Drexler JF, Corman VM, Müller MA, Maganga GD, Vallo P, Binger T, Gloza-Rausch F, Rasche A, Yordanov S, Seebens A, Oppong S, Adu Sarkodie Y, Pongombo C, Lukashev AN, Schmidt-Chanasit J, Stöcker A, Carneiro AJ, Erbar S, Maisner A, Fronhoffs F, Buettner R, Kalko EK, Kruppa T, Franke CR, Kallies R, Yandoko ER, Herrler G, Reusken C, Hassanin A, Krüger DH, Matthee S, Ulrich RG, Leroy EM & Drosten C. (2012). Bats host major mammalian paramyxoviruses. Nat. Commun., 3, 796. <Law: 10.1038/ncomms1796>.
[7] Sulkin SE & Allen R. (1974). Virology. Vol. 8, Basel: Melnick JL, Karger.
[8] Luis AD, Hayman DT, O’Shea TJ, Cryan PM, Gilbert AT, Pulliam JR, Mills JN, Timonin ME, Willis CK, Cunningham AA, Fooks AR, Rupprecht CE, Wood JL & Webb CT. (2013). A comparison of bats and rodents as reservoirs of zoonotic viruses: are bats special? Proc. Biol. Sci. 280 (1756), 20122753. <Law: 10.1098/rspb.2012.2753>.
[9] Olival KJ, Hosseini PR, Zambrana-Torrelio C, Ross N, Bogich TL & Daszak P. (2017). Host and viral traits predict zoonotic spillover from mammals. Nature, 546 (7660), 646-650. <doi: 10.1038/nature22975>.
[10] Anthony SJ, Johnson CK, Greig DJ, Kramer S, Che X, Wells H, Hicks AL, Joly DO, Wolfe ND, Daszak P, Karesh W, Lipkin WI, Morse SS; PREDICT Consortium, Mazet JAK & Goldstein T. (2017), Global patterns in coronavirus diversity. Virus Evol. 3 (1), vex012. <Law: 10.1093/ve/vex012>.
[11] Letko M, Seifert SN, Olival KJ, Plowright RK & Munster VJ. (2020). Bat-borne virus diversity, spillover and emergence. Nat. Rev. Microbiol. 18 (8), 461-471. <doi: 10.1038/s41579-020-0394-z.>.
[12] Mollentze N & Streicker DG. (2020). Viral Zoonotic Risk Is Homogenous Among Taxonomic Orders of Mammalian and Avian Reservoir Hosts. Proc. Natl. Acad. Sci. USA, 117 (17), 9423-9430. <doi: 10.1073/pnas.1919176117>.
[13] Jebb D, Huang Z, Pippel M, Hughes GM, Lavrichenko K, Devanna P, Winkler S, Jermiin LS, Skirmuntt EC, Katzourakis A, Burkitt-Gray L, Ray DA, Sullivan KAM, Roscito JG, Kirilenko BG, Dávalos LM, Corthals AP, Power ML, Jones G, Ransome RD, Dechmann DKN, Locatelli AG, Puechmaille SJ, Fedrigo O, Jarvis ED, Hiller M , Vernes SC, Myers EW & Teeling EC. (2020). Six reference-quality genomes reveal evolution of bat adaptations. Nature, 583, 579-584. <doi: 10.1038/s41586-020-2486-3>.
[14] Zhang T, Wu Q & Zhang Z. (2020). Probable Pangolin Origin of SARS-CoV 2 Associated with the COVID-19 Outbreak. Curr. Biol. 30 (7), 1346-1351.e2. <doi: 10.1016/j.cub.2020.03.022>.
[15] Damas J, Hughes GM, Keough KC, Painter CA, Persky NS, Corbo M, Hiller M, Koepfli KP, Pfenning AR, Zhao H, Genereux DP, Swofford R, Pollard KS, Ryder OA, Nweeia MT, Lindblad-Toh K, Teeling EC, Karlsson EK & Lewin HA. (2020). Broad host range of SARS-CoV-2 predicted by comparative and structural analysis of ACE2 in vertebrates. Proc. Natl. Acad. Sci U.S.A. 2020 Aug 21:202010146. <doi: 10.1073/pnas.2010146117>.
[16] O’Shea TJ, Cryan PM, Cunningham AA, Fooks AR, Hayman DTS, Luis AD, Peel AJ, Plowright RK & Wood JLN. (2014). Bat flight and zoonotic viruses. Emerg. Infect. Dis. 20 (5): 741-745. <doi:10.3201/eid2005.130539>.
[17] Brook CE & Dobson AP. (2015). Bats as special reservoirs for emerging zoonotic pathogens. Trends Microbiol. 23 (3), 172-180. <doi: 10.1016/j.tim.2014.12.004>.
[18] Zhang G, Cowled C, Shi Z, Huang Z, Bishop-Lilly KA, Fang X, Wynne JW, Xiong Z, Baker ML, Zhao W, Tachedjian M, Zhu Y, Zhou P, Jiang X, Ng J, Yang L, Wu L Xiao J, Feng Y, Chen Y, Sun X, Zhang Y, Marsh GA, Crameri G, Broder CC, Frey KG, Wang L-F & Wang J. (2013). Comparative analysis of bat genomes provides insight into the evolution of flight and immunity. Science, 339 (6118), 456-460. <doi: 10 .1126/science.1230835>.
[19] Banerjee A, Baker ML, Kulcsar K & Misra V, Plowright R, Mossman K. (2020). Novel insights into immune systems of bats. Front. Immunol. 11, 26. <doi: 10 .3389/fimmu.2020.00026>.
[20] Foley NM, Hughes GM, Huang Z, Clarke M, Jebb D, Whelan CV, Petit EJ, Touzalin F, Farcy O, Jones G, Ransome RD, Kacprzyk J, O’Connell MJ, Kerth G, Rebelo H, Rodrigues L, Puechmaille SJ & Teeling EC. (2018). Growing old, yet staying young: The role of telomeres in bats’ exceptional longevity. Sci. Adv. 4 (2), eaao0926. <10.1126/sciadv.aao0926>.
[21] Kacprzyk J, Hughes GM, Palsson-McDermott EM, Quinn SR, Puechmaille SJ, O’Neill LAJ & Teeling EC. (2017). A potent anti-inflammatory response in bat macrophages may be linked to extended longevity and viral tolerance. Acta Chiropter, 19 (2), 219-228. <doi: 10.3161/15081109ACC2017.19.2.001>.
[22] Mandle JN, Schneider C, Schneider DS, Baker ML. (2018). Going to bat(s) for studies of disease tolerance. Front Immunol, 9 (2112). < doi: 10.3389/fimmu.2018.02112>.
[23] Gibb R, Redding DW, Chin KQ, Donnelly CA, Blackburn TM, Newbold T & Jones KE. (2020). Zoonotic host diversity increases in human-dominated ecosystems. Nature, 584 (7821), 398-402. <doi: 10.1038/s41586-020-2562-8>.
[24] Afelt A, Frutos R & Devaux C. (2018). Bats, Coronaviruses, and Deforestation: Toward the Emergence of Novel Infectious Diseases? Front. Microbiol. 9, 702. <doi: 10.3389/fmicb.2018.00702>.
The Encyclopedia of the Environment by the Association des Encyclopédies de l'Environnement et de l'Énergie (www.a3e.fr), contractually linked to the University of Grenoble Alpes and Grenoble INP, and sponsored by the French Academy of Sciences.
To cite this article: LARCHER Gérald (January 5, 2025), Bats and viruses or how to live together in harmony, Encyclopedia of the Environment, Accessed May 24, 2026 [online ISSN 2555-0950] url : https://www.encyclopedie-environnement.org/en/life/bats-viruses-how-live-together-harmony-2/.
The articles in the Encyclopedia of the Environment are made available under the terms of the Creative Commons BY-NC-SA license, which authorizes reproduction subject to: citing the source, not making commercial use of them, sharing identical initial conditions, reproducing at each reuse or distribution the mention of this Creative Commons BY-NC-SA license.




