The marine environment
PDF
More than two thirds of the Earth’s surface is occupied by seas and oceans. Heated by the sunshine, the temperature of sea water varies with location and depth. On the other hand, its composition and therefore its salinity are remarkably constant in a given sea while showing some variations from one sea to another. In the marine environment, the cradle of life, under the open ocean surface, which is neither flat as the ancients believed, nor really round, a multitude of organisms live in conditions very different from those known on earth and constitute an amazing biodiversity.
1. Origin and extent of the oceans
During the slow evolution of our planet, plate tectonics has continuously changed the positions of continents and oceans. At the time of the Pangea, about 300 million years ago [1], there was only one large ocean surrounding this one continent. Today, according to the classification of the International Hydrological Organization (IHO), there are three oceans. The Pacific Ocean is the largest, its surface area is about half that of the oceans as a whole, and it alone covers one-third of the Earth’s surface. It is certainly because of its predominance on the surface that the median meridian of this ocean was chosen as the date change line. The Atlantic Ocean is the second largest by area, accounting for about 30% of the total. It is much better supplied with fresh water than other oceans, since it receives flows from large rivers such as the Amazon, Congo and St. Lawrence. The Indian Ocean, the third largest by area, accounts for about 20% of the total. It is almost entirely located in the southern hemisphere, between Asia, Africa and Australia.
Despite this official classification, it is common practice to have 5 oceans on our planet, rather than 3, distinguishing between the Antarctic Ocean to the south, or Austral, which surrounds the Antarctic continent up to about 60 degrees and whose area represents about 6% of the total, and the Arctic Ocean to the north, which is bordered by the lands of Siberia, Scandinavia, Greenland and North America, whose area represents about 4% of the total. This shallow Arctic Ocean is partly covered by ice floes. Globally, the surface area of these oceans represents 71% of that of the globe.
The seas then appear as marine sub-domains, of relatively small sizes and relatively individualized. Geographical reasons usually justify this individualisation, as is the case for the Mediterranean Sea, which communicates with the Atlantic Ocean only through the Strait of Gibraltar, so narrow that the Atlantic tides are slowed down to the point of being felt very little on the Mediterranean coast (read The Tides). On the contrary, the English Channel is largely open to the Atlantic and receives high tides, slowed by the Pas de Calais between England and France, before joining those of the North Sea, being slowed down again by the narrow passages between Denmark and Sweden, and finally reaching the Baltic Sea almost imperceptible. In other cases, such as that of the Caribbean Sea, even if the geographical separation is very real and characterized by the long chain of islands that runs from the coasts of Venezuela to the Florida peninsula, it is rather because of the history of the riparian states and successive conquests that we can justify the individualization of these seas.
2. Density, salinity and temperature of the marine environment
Under reference conditions (pressure of 1013 hPa, or hectoPascal, temperature of 3.98 °C, zero salinity) the density of the water is exactly 103 kg/m3. It was these conditions that led to the first definition of the kilogram, the maximum value of the mass of a litre of fresh water. The density of seawater varies mainly with temperature and salinity, much less with pressure, which often leads to considering this fluid as incompressible.
The salinity, or mass fraction of salt, or ratio of the mass of salt contained in a unit of volume to the mass of this unit, is expressed in g/kg (gram of salt per kilogram of sea water). In lakes and rivers, salinity is almost zero, rarely exceeding a few units. It can reach and sometimes exceed 50 g/kg in the seas, its average value is around 35 g/kg. It is 12 g/kg in the Black Sea. In the Dead Sea, its very high value, close to 275 g/kg, practically prohibits any animal or plant life.

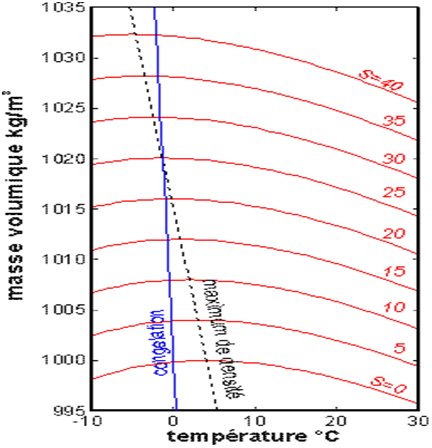
The variation in density as a function of temperature, shown in Figure 2, shows its maximum, around 3.98°C for fresh water, with a decreasing variation between 13 and 30°C. In addition, in summer, the surface water of the warmest seas can reach temperatures of 26 to 30°C, which often leads to cyclones (see Tropical Cyclones, Development and Organisation).
In the upper layers of the marine environment, surface water, heated by solar radiation, is subjected to constant thermal exchanges by conduction and convection with the atmosphere. Agitation by waves and turbulence then manages to homogenize the temperature in the first tens of meters (between 0 and -50 m). On the contrary, at great depths (below -120 m), exchanges are almost limited to pure conduction and become much weaker, so that the approximation of a resting marine environment is well justified, even if extremely slow deep currents, coupled with surface currents, weakly amplify the apparent thermal conductivity of the water at the depths.
Between these two areas, it is common practice to distinguish a relatively thin layer (between -50 and -120 m) called the thermocline, where the temperature can vary by about ten degrees between the water above and the water below. The temperature of the water above the thermocline experiences significant seasonal variations, due to variations in sunlight, without any change in the temperature of the deep layers. It can be seen in Figure 1 that the Mediterranean Sea thermocline is built in spring, as the sunlight increases. It reaches its maximum temperature difference of around 10°C in August, and sees it decrease more and more in autumn and early winter. In February and March, the thermocline disappears, making the temperature almost invariant over the entire depth, between 13 and 13.5 °C. It is noteworthy that above and below the thermocline the temperature variation as a function of altitude, even very low, remains increasing, so that this deep fluid layer heated above is only slightly subject to the convective instability of Rayleigh-Bénard, which generates a low rise in deep waters compensated by a fall in surface waters.
However, this vertical movement plays a key role in the deep life of the marine environment by regenerating oxygen by supplying surface water. The corresponding upwelling is beneficial to life on the surface by bringing nutrients from the seabed to the surface. In the Mediterranean, this phenomenon of deep convection is mainly located in the Gulf of Lions, making it a major centre of biological activity. However, this convection only occurs when the conditions necessary for its presence are met, so the Black Sea remains constantly stratified in density, and this prevents the penetration of oxygen beyond a depth of 200 m. Only very specific species can live in this marine environment under so-called anoxic conditions. On the global scale of thermohaline circulation a similar phenomenon of convection brews the world ocean at depth.
3. Composition of seawater
The chemical composition of seawater is not a simple matter. Most of the chemical elements are found in solution in the form of a complex mixture of anions, cations and molecules. Ions are not reduced to either chloride anion or sodium cation, although these two elements, which are largely dominant, form the basis of sea salt (NaCl). The attached Table lists, in descending order of importance, the top five anions and cations of typical seawater, with a salinity of 35 g/kg. As it happens, the ratios between the concentrations of all these ions vary very little from sea to sea [3], so that, if we measure the content of one of these constituents, we can deduce the overall salinity.
These elements have various origins. Some ions come from the dissolution of continental rocks by rivers that carry them to the oceans, where they stay for very long periods of time and where evaporation of water increases their concentration. A significant part of the cations comes from the original ocean floor. And the origin of the chloride ion is often attributed to the degassing of hydrogen chloride from volcanoes, which is soluble in water.
Table 1. Concentrations of the main ions dissolved in typical seawater with a salinity of 35 g/kg.
In addition to water and salts, there are also various low-concentration molecules, such as boric acid (0.0198 g/kg) and carbon dioxide (0.0004 g/kg), as well as nitrogen and oxygen. It is remarkable that the amount of carbon dioxide in seawater is much greater than in the air, about 60 times, without this value constituting an upper limit on the oceans’ ability to retain this molecule. This is a highly debated issue at the moment: the possibility of sequestering carbon dioxide in the oceans, with a view to reducing the content of this greenhouse gas in the atmosphere. Some suggest capturing this gas near emitting sources and injecting it directly into the oceans at great depth, despite uncertainties about possible reactions, such as significant changes in the pH [4] of seawater or the carbon cycle.
4. The hummocky relief of the seas
Since mass is not uniformly distributed in the Earth’s mantle, gravity cannot be uniform on the Earth’s surface. This effect alone implies that the altitude of oceans assumed to be absolutely immobile must vary due to the opposite of local gravity, being maximum where gravity is minimal, and vice versa, so that their product is constant. As a result, the average free surface of the oceans cannot coincide exactly with that of a sphere. However, since all altitudes are defined and measured from mean sea level, it is necessary to specify what this level represents [5]. This has led oceanographers and geophysicists to choose as a reference the equipotential surface of the gravity field (or gravity) that best coincides with the mean sea level. This particular surface, called the geoid, shown in Figure 3, highlights how far the actual ocean surface deviates from the sphere that would correspond to the case of uniform gravity. This bumpy surface represents the variations in gravity on the Earth’s surface, both on continents and oceans [6].
5. The amazing biodiversity of the marine environment

References and notes
[1] The single continent called Pangea dates back to the Paleozoic (between 500 and 250 million years ago, or Ma, before our time), a geological era that followed the Precambrian (2500 to 500 Ma) and preceded the Mesozoic (250 to 65 Ma), the Cenozoic (65 to 1.65 Ma) and the present Quaternary era.
[2] S. Levitus and T.P. Boyer, 1994, World Ocean Atlas, Vol. 4, Temperature, NOAA
[3] This empirical property is known as Dittmar’s Law, in honour of the Scottish chemist William Dittmar (1859-1951) who drew it from the analyses of samples taken during the Challenger oceanographic expedition between 1872 and 1876.
[4] The acronym pH stands for hydrogen potential. Its value measures the chemical activity of hydrogen ions (H+) in solution and characterizes the acidity or basicity of a solution. Thus, in an aqueous medium at 25°C, a solution is acidic with a pH below 7, neutral with a pH of 7 and basic with a pH above 7.
[5] Gravity, or the weight of the unit of mass, derives from a potential. Like all vector quantities that possess this property, it can be represented by a family of equipotential surfaces to which it is orthogonal. These surfaces are tighter, like contours on a map, where gravity is at its highest; they are further apart where gravity is at its lowest. Broadly speaking, we can consider that the bumps of the geoid correspond to gravity minima, the hollows to maxima.
[6] G. Balmino, F. Perosanz, R. Rummel, N. Sneeuw and H. Sunkel, Champ, GRACE and GOCE: mission concepts and simulations, Int. Gravity Commission and Int. Geoid Commission, N°2, Trieste, 1999, vol. 40, No. 3-4, pp. 309-319.
The Encyclopedia of the Environment by the Association des Encyclopédies de l'Environnement et de l'Énergie (www.a3e.fr), contractually linked to the University of Grenoble Alpes and Grenoble INP, and sponsored by the French Academy of Sciences.
To cite this article: MOREAU René (January 5, 2025), The marine environment, Encyclopedia of the Environment, Accessed May 22, 2026 [online ISSN 2555-0950] url : https://www.encyclopedie-environnement.org/en/water/marine-environment-2/.
The articles in the Encyclopedia of the Environment are made available under the terms of the Creative Commons BY-NC-SA license, which authorizes reproduction subject to: citing the source, not making commercial use of them, sharing identical initial conditions, reproducing at each reuse or distribution the mention of this Creative Commons BY-NC-SA license.








