Forests facing global environmental change
PDF
Millennia of large-scale land clearing and intensive logging have significantly reduced the area of forest in France. Despite reforestation efforts dating back to the 19th century, human activities have placed significant pressure on trees and forests. Since the Industrial Revolution, air pollution and rising levels of greenhouse gases in the atmosphere have triggered a climate change that is intensifying. Trees are suffering the consequences of toxic pollutants, intense or repeated droughts, high temperatures and storms. This results in a significant reduction in their growth, marked defoliation, a lack of branches and increased mortality. The weakening of trees’ natural defences then exposes them to biotic attacks, such as insects, fungi or other pathogens. These attacks exacerbate the decline, causing long-term damage to the health and resilience of forest stands.
1. The impact of human activities on forests
Human activities on Earth have led to (i) massive environmental pollution (air, water, soil), (ii) the climate change largely due to the continuous increase in atmospheric CO2 concentrations [1], and (iii) serious damage to biodiversity.
Thus, since the beginning of the Industrial Revolution, the CO2 content in the atmosphere has increased by 50%, rising from 280 ppm in 1800 to 424 ppm in 2024. In particular, this results into an inevitable rise in the Earth’s average global earth temperature of approximately 1.5°C to 1.6°C between 1850 and 2024, exacerbating extreme events (droughts, heatwaves, etc.), impacting all ecosystems, particularly forests, as well as plant, animal and human health.
Yet as early as 1800, Alexander von Humboldt, followed by other pioneers of ecology in the 19th century, warned of the effects of deforestation on the climate and advocated for a harmonious relationship between humans, nature, trees and forests (see Focus Some Pioneers of Ecology).
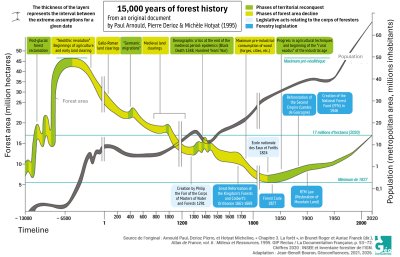
Following the last ice age in Europe, the area covered by forest had increased significantly thanks to a more favourable climate (Figure 1). Then, from the onset of agriculture (around 6000 BC), forest cover in France steadily declined until the 19th century due to extensive felling and slash-and-burn practices, despite some temporary recoveries (the fall of the Roman Empire, the Hundred Years’ War, the Black Death – Figure 1) [2]. Colbert’s 1669 ordinance on forests only temporarily stabilised the situation, but proactive planting and conservation efforts led to a marked increase in forest cover until the mid-20th century.
After the Second World War, reforestation by the National Forest Fund and the widespread abandonment of agricultural and pastoral land, which reverted to woodland naturally, accelerated the return of forests, a process that has continued uninterrupted into the 21st century.
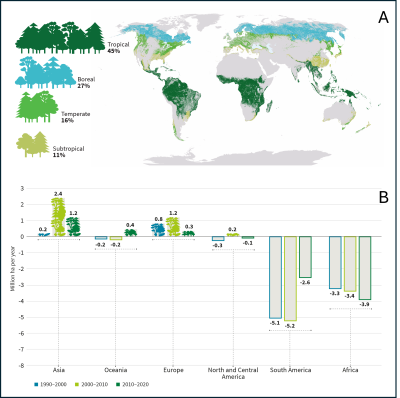
- A sharp decline in tropical forest area has been observed in South America and Africa over the past three decades (Figure 2B), mainly due to forest fires and deforestation in the Brazilian Amazon and the Democratic Republic of the Congo for agricultural purposes (subsistence or industrial, soya, etc.), and timber harvesting.
- In Asia, Indonesia is actively contributing to this decline by replacing its forests with oil palm plantations.
These deliberate acts of deforestation account for 58% of global CO₂ emissions into the atmosphere.
In Europe, improved management linked to a proactive reforestation policy had led to a significant increase in forest cover between 1990 and 2010, and with it carbon storage, but this growth slowed during the decade from 2010 to 2020 (only +0.3 million ha compared with 1.2 million ha previously, Figure 2B).
2. Current factors contributing to forest decline
The combined effects of droughts, heatwaves, storms [4] and diseases reduce tree growth and increase tree mortality. Forest dieback linked to severe droughts, which has been observed on every continent since the 19th century, has become more frequent and widespread in recent decades [5],[6].
This effect is exacerbated by forest fires (whether deliberate or caused by climatic conditions that encourage their outbreak), which are devastating across the globe: (i) in tropical regions (the Amazon, etc.), (ii) in North America (the USA, Canada), (iii) in Europe (France, Italy, Spain, Portugal, Greece) [7], (iv) in boreal regions (Russia, etc.) (v) and also in Australia and Indonesia.
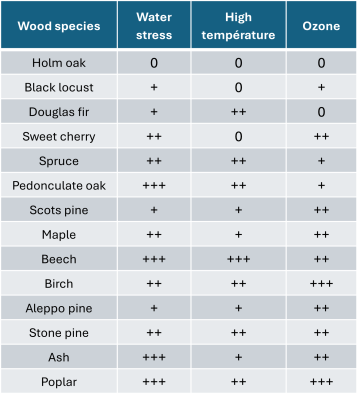
In addition to all these phenomena, there is also atmospheric pollution. The most common European forest species exhibit varying degrees of sensitivity to a whole range of stresses (Table 1). We must also consider biotic stresses (fungi, insects, bacteria), which are more damaging to trees already weakened by climate change (see section 5).
Table 1. Sensitivity of various forest species to various stresses. Adapted from Claessens H, 2016[8]. 0: tolerant; +:not very sensitive; ++: sensitive; +++: very sensitive.
The consequence of this decline is that forests, which are major carbon sinks, are absorbing less CO₂. Their storage capacity fell by around 30% between 1990 and 2010 [9]. In the Amazon, this decline is thought to have occurred in just 20 years, between 2000 and 2010 [9]. Forest fires, by emitting massive amounts of CO2 [10], weaken the forest carbon sink, creating a vicious circle (See Understanding and preventing wildfires).
In France, annual carbon sequestration has fallen from 63 million tonnes (Mt) of CO₂ (2005–2013) to 39 Mt (2014–2022) according to the latest forest inventories [9].
Massive tropical deforestation, combined with increasing climate impacts (droughts, fires, tree mortality), is thus severely undermining the role of forests as carbon sinks on a global scale, including in Europe and France, where resilience is declining.
3. Trees and pollution

Ever since humans settled in one place, the use of fire for heating homes and clearing land has led to air pollution, a problem that was greatly exacerbated by the industrial revolutions from the mid-18th century onwards (see Focus Air Pollution and trees).
Until the 1980s, air pollution was dominated by SO₂ resulting from the burning of coal in factories and homes. Ozone then became the predominant pollutant (see Air Pollution). This secondary pollutant is formed under the influence of ultraviolet (UV) rays from nitrogen oxides, hydrocarbon residues and volatile organic compounds (VOCs) originating from road traffic, but also from certain plants, particularly during periods of high heat and drought. These pollutants have been shown to have harmful effects on all plants, attacking their leaves and causing losses in biomass[11],[12],[13] (See What is the impact of air pollutants on vegetation?).
The visible effects of ozone on trees are observed in the Vosges Mountains when easterly winds carry polluted air from the Rhine plains. A marked yellowing appears on the needles of spruce trees (Figure 3A). In Greece, Aleppo pine needles display spots typical of ozone damage, known as ‘chlorotic mottling’ (Figure 3B).
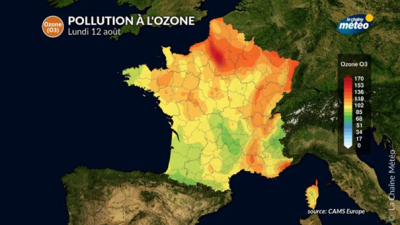
The vulnerability of forests stems from the interaction between ozone and the effects of climate change [14]. A recent study shows that in the Amazon, drought-related fires exacerbate the damage caused by ozone, the formation of which is promoted by nitrogen oxides released from the burning of trees [10]. The result is that there is more CO₂ in the atmosphere, both from emissions from the fires and because ozone reduces photosynthesis.
4. Forests in the face of droughts and heatwaves
4.1 Physiological responses of trees to drought and high temperatures
Global warming (i) extends the growing season by several days per decade, with (ii) earlier budburst and (iii) delayed senescence. This increases trees’ water consumption and heightens the risk of water stress. Episodes of drought and heatwaves are also becoming more frequent.
Water flows from the roots to the leaves thanks to a water potential gradient (see The tireless-quest for water by plants). An oak tree with a diameter of 45 cm at breast height can thus absorb around a hundred litres a day. When the soil dries out, this flow can be interrupted, causing cavitation (air bubbles) in the vessels. Before it comes to this, the stomata, which regulate transpiration from the leaves, close to prevent excessive water loss. But in doing so, they reduce the intake of CO₂, the fuel for biochemical reactions during photosynthesis (See The path of carbon in photosynthesis): the tree’s growth is thus slowed down.
Temperature plays a key role in photosynthesis, with each type of forest having its own optimal temperature (See Effects of temperature on photosynthesis):
- Boreal forests: optimum around 20 °C;
- Temperate forests: optimum around 25–30 °C;
- Tropical forests: optimum around 30–35 °C (often close to 30 °C for the canopy).
When the temperature exceeds these optimum levels, (i) photosynthetic enzymes are impaired, (ii) chlorophyll pigments degrade, all of which causes the leaves or needles to discolour, then dry out and fall.
Finally, it is becoming increasingly difficult to distinguish the effects of high temperatures from those of droughts, as these two phenomena very often occur together.
4.2 Current impact of droughts and high temperatures on forests
Forests have been affected by droughts for decades, both in France and in neighbouring countries [15],[16]. Significant episodes have occurred historically (e.g. 1892–1897, 1910–1917, 1922–1927, 1946–1949, 1955–1961, 1976, 1989–1991). Their frequency has been increasing since the end of the 20th century, often in combination with intense heatwaves.

- In the French Mediterranean region, the average rainfall deficit for 2003–2007 (-48% over five years) remains a record for the last 160 years.
- In Provence, eight of the last ten years (2015–2024) have been both water-deficient and very hot, exacerbating defoliation that has been rising steadily since 1990: 52% for deciduous trees and 42% for conifers in 2024 (Figure 5).
In France, rates of defoliation and tree mortality (Figure 6) have been rising since 2003, reducing forest productivity, particularly at the southern limits of species’ ranges or at low altitudes for mountain species [18]. This has also been observed at the heart of species’ ranges during exceptional years. As in north-eastern France following 2019, a year marked by a rainfall deficit between January and September, coupled with a very high average temperature (the third warmest year on record), two heatwaves breaking all-time records and exceptional sunshine levels (+10 to 20%).
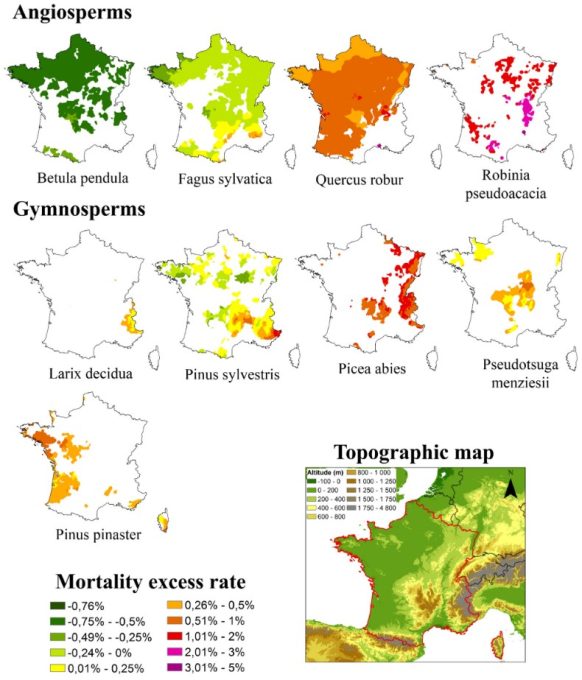

- For two dominant pine species in the French Mediterranean region, Scots pine and Aleppo pine [19],[20], it declined sharply at the turn of the 21st century (Figure 7):
- The Aleppo pine (the most drought-resistant native species) initially benefited from warming in the 20th century, before a sharp reversal; its average defoliation reached 40% in Provence in 2019 (climate stress + opportunistic pathogens).
- The Scots pine, at the lower limit of its range, has been declining rapidly for over a century.
- The downy oak and the holm oak saw their productivity increase by more than 40% during the 20th century, but they have been dying off on a massive scale since 2003–2007:
- Two-thirds of these oaks have a defoliation rate of over 50%;
- The lower limit of healthy downy oak has shifted several hundred metres higher in altitude and around 100 km further.
Paradoxically, species reputed to be resistant to heat or drought (Aleppo pine, holm oak, and the dominant shrubs of the garrigue) are suffering severe dieback, even though the climate has not exceeded their known biological limits. Various explanations come into play (see Chapter 4.2 and forthcoming article ‘Forests and climate change: risks and solutions’): the age of the stands, genetic factors, soil degradation leading to declining fertility, systemic disruptions to ecosystems, and pathogen-induced upheavals.
The repeated occurrence of stress over several years prevents recovery, thereby reducing resilience. A good example is provided by the high mortality rates and health deterioration in the Provençal cork oak forests (suberayes) following repeated bouts of fire and drought.
Resilient immediately after the fires and the 2003 heatwave, which followed several wet years, these forests suffered severe dieback from 2005–2007 onwards, which was exacerbated at sites that had experienced several fires in the preceding 50 years. The fires of 2007, which occurred during the fifth consecutive dry year, devastated stands even in areas that had not burned for 50 years. By 2025, due to repeated droughts over the previous decade, this forest ecosystem had still not recovered, with high mortality rates and a severely degraded overall health [21].
This vicious cycle affects the entire ecosystem (flora, soil, fauna, microorganisms): repeated droughts make ecosystems much more susceptible to fire, and repeated fires make them much more susceptible to droughts and heatwaves. At the national level in France, INRAE projections [22] indicate:
- An increase in fire activity from 2030 onwards (+13 to +22% depending on the indicators: area burnt, out-of-control fires, major fires).
- An increase in the number of large wildfires (>100 ha), which could rise from around 7 per year to 10 by 2050 and as many as 20 by 2090 under the worst-case scenario.
Globally, a fourfold increase in large wildfires is expected by the end of the century, exacerbated by the increased frequency and duration of droughts and heatwaves, posing a long-term threat to forest ecosystems.
5. Interactions between constraints
The decline of a forest species is rarely caused by a single environmental factor. As a multifactorial process, it often results from the simultaneous or successive effects of several abiotic and biotic factors, whose complex interactions lead to their decline [23].
A healthy tree produces defence molecules that are effective against the various attacks it usually faces (See Environmental constraints and oxidative stress in plants). However, when it is subjected to primary abiotic stress (drought, pollution, etc.), its defences weaken, making it vulnerable to a secondary attack by a pathogen or pest. Conversely, a tree already weakened by a parasite or disease will be more susceptible to abiotic stresses (Figure 8) [16]. Finally, abiotic stresses themselves can interact, such as fires and droughts, or droughts and heatwaves.
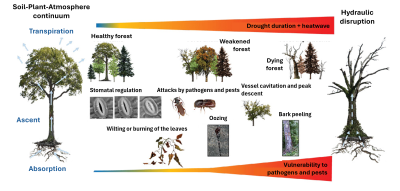
5.1 Wilting caused by abiotic stress


Recent mortality of Aleppo pine in the Mediterranean. Mortality of Aleppo pine is exacerbated by recurrent droughts and is correlated with high ozone concentrations in Mediterranean forests [25]. Attacks by bark beetles (small beetles, see 4.2 and Focus Biotic pests and forest decline) between 2015 and 2017 accelerated this decline.
Spruce dieback in Central and Eastern Europe. Over the past decade or so, spruce trees weakened by drought and heatwaves have suffered high mortality rates due to bark beetle infestations (Figure 10), particularly in eastern France and Central Europe. Between 2018 and 2024, the volume of deadwood in Europe is expected to reach around 250 million m³, exceeding the damage caused by the storms of 1999 (200 million m³ in Europe, including 140 million in France).
Wider ecosystem impacts. Primary stresses disrupt the entire forest ecosystem, including predator-prey balances, trophic interactions and symbioses, particularly in the soil (see article in preparation ‘Forests and climate change: risks and solutions’). This leads to a loss of soil fertility and biodiversity, making trees more vulnerable. In the long term, the decline in key species such as seed dispersers and pollinators, due to adverse climatic conditions, prevents the replacement of dead trees and exacerbates forest decline.
5.2 Increase and intensification of biotic stresses
Global warming greatly favours biotic pests, which often become the dominant factors in forest decline (see Focus: ‘Biotic pests and forest decline’).
- Wood-boring and defoliating insects
Bark beetles, small wood-boring beetles that tunnel beneath the bark of trees play a major role in forest decline. Global warming allows them to complete up to two additional reproductive cycles per year, causing their population to grow exponentially [27] and extending their attacks over several months. The resulting tree mortality provides further opportunities for these insects to proliferate, creating a vicious circle (see Focus Biotic pests and forest decline).
Another example is that of the processionary caterpillar, which is linked to the decline of Scots pine in south-eastern France [28]: the significant decline in winter cold has provided it with both a high survival rate and an extended period of activity. Over 50 years, its population density has increased, and it has spread 300 km in latitude and 300 metres in altitude.
- Seed predators and compromised reproduction
The situation is paradoxical for beech trees (and for certain oak species), whose reproductive strategy had previously been concentrated in years spaced far apart. Global warming is making their fruiting more regular, thereby maintaining high population s of predators that feed on their seeds. This increased predation consequently jeopardises their reproduction [29].
- Fungal diseases favoured by longer growing seasons
In temperate and cold climates, pathogenic fungi now benefit from significantly longer periods of proliferation (early spring and late autumn). Aleppo pine shoot canker, a low-level endemic in the 20th century, has become epidemic. It ravages the shoots of this species and severely limits its growth.
- Invasive pathogens and pests (globalisation)
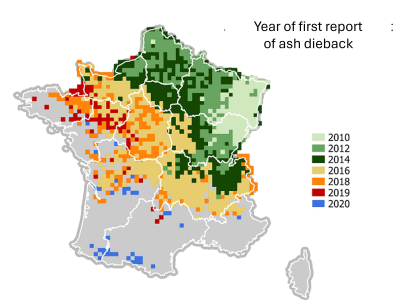
With globalisation, the accidental introduction of diseases and pests is causing widespread devastation. A prime example is ash dieback, a disease caused by the fungus Chalara fraxinea. Imported from Asia, it reached Western Europe via Poland. It is decimating French ash populations, spreading very rapidly since its first appearance in 2010 (Figure 12).
The decline of forest species is therefore a complex phenomenon linked to the interactions between numerous climatic, abiotic, biotic and human factors. Each case remains unique (species, location within the range, silvicultural history, soil, management, etc.). There is no one-size-fits-all solution, but appropriate silvicultural approaches can significantly enhance the resistance and resilience of most stands to current dominant stresses (article in preparation: ‘Forests facing climate change: risks and solutions’).
6. Messages to remember
- Forest destruction and decline began very early in human history, particularly with the settlement of populations and systematic land clearing, which led to a sharp reduction in forested areas.
- The Industrial Revolution marked the beginning of a gradual decline in trees and forests, linked to increasing human pressures (pollution, changes in land use, etc.).
- In recent decades (particularly since the 1990s and 2000s), this phenomenon has become much more pronounced, in parallel with the acceleration of climate change. Trees are increasingly subjected to combined episodes of prolonged drought, heatwaves and ozone pollution.
- Climatic stresses weaken trees’ natural defences, making them much more vulnerable to attacks by insects, fungi,
- Conversely, increasingly severe and frequent biotic attacks (fuelled by global warming and globalisation) are making trees more vulnerable to climatic We are thus witnessing a veritable downward spiral: the climate predisposes and triggers the problem, pests exacerbate and accelerate the decline, whilst repeated fires and droughts mutually amplify their negative effects.
Notes et references
- Cover image. Waldsterben [Photo Colin Behrens, via Pixabay]
[1] IPCC, 2021: Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [Masson-Delmotte, V et al.)]. DOI officiel : https://doi.org/10.1017/9781009157896, Lien direct vers la page IPCC : https://www.ipcc.ch/report/ar6/wg1/
[2] Dodane C., « La forêt ardéchoise : une ressource territoriale à révéler et à gérer, une nouvelle organisation de l’espace », Géoconfluences, octobre 2010. – https://geoconfluences.ens-lyon.fr/doc/territ/FranceMut/FranceMutScient8b.htm – Arnould P, Derioz P & Hotyat M (1995). Chapitre 3 : La forêt, pp 53-72. Dans Brunet R & Auriac F. Atlas de France, vol. 6 : Milieux et Ressources, Paris. GIP Reclus/ La Documentation Française. Chiffres 2020 : INSEE et Inventaire forestier de l’IGN. Adaptation : Jean-Benoît Bouron, Géoconfluences, 2021, 2026.
[3] FAO. 2020. Évaluation des ressources forestières mondiales 2020 – Principaux résultats. Rome. https://doi.org/10.4060/ca8753fr
[4] A côté des tempêtes Lothar et Martin en décembre 1999 qui ont dévasté près de 1 million d’ha de forêts françaises, les tempêtes Nils et Pedro de février 2026 ont couché les conifères (pins et épicéas) du massif d’Espinouse dans l’Hérault.
[5] Hammond WM et al. (2022). Global field observations of tree die-off reveal hotter-drought fingerprint for Earth’s forests. Nature Communications, 13, 1761.
[6] Allen CD et al. (2010). A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. Forest Ecology and Management, 259, 660-684.
[7] Vennetier M. (2006). Pourquoi les forêts brûlent-t-elles ? Edition Le Pommier, 64p.
[8] Claessens H (2016). Quelques considérations pour adapter nos forêts aux changements climatiques. Silva Belgica, Janvier-Février 2016, 20-29.
[9] Mirabel A, Derroire G, Sist P & Traissac S (2025). Comment une forêt peut-elle émettre plus de CO2 qu’elle n’en capture ? The Conversation, 2/10/2025.
[10] Brown F, Sitch S, Folberth G, Cheesman A, Mercado L & Barningham S (2024). Fire activity and drought increase ozone-plant damage to the Amazon rainforest, Physical Sciences. https://doi.org/10.21203/rs.3.rs-4802855/v1.
[11] Wittig VE, Ainsworth EA, Naidu SL, Karnosky DF & Long SP (2009). Quantifying the impact of current and future tropospheric ozone on tree biomass, growth, physiology and biochemistry: a quantitative meta-analysis. Global Change Biology, 15, 396–424, doi: 10.1111/j.1365-2486.2008.01774.x
[12] Dizengremel P (2021). L’ozone et les plantes, histoire d’une relation toxique. The Conversation, 2 mai 2021
[13] Ferretti M et al. (2024). The fingerprint of tropospheric ozone on broadleaved forest vegetation in Europe. Ecological Indicators, 158, 111486.
[14] De Marco A et al. (2022). Strategic roadmap to assess forest vulnerability under air pollution and climate change. Global Change Biology, 28, 5062-5085. DOI: 10.1111/gcb.16278
[15] Bréda N, Huc R, Granier A & Dreyer E (2006). Temperate forest trees and stands under severe drought: a review of ecophysiological responses, adaptation processes and long-term consequences. Annals of Forest Science, 63, 625-644.https://doi.org/10.1051/forest:2006042
[16] Vitasse Y, Wohlgemuth T & Rigling A (2023). Les forêts face aux sécheresses et canicules : causes de dépérissements, facteurs aggravants et différences de sensibilité entre les espèces. Revue forestière française, 74, 121-132. https://doi.org/10.20870/revforfr.2023.7586
[17] Lu R, Williams L.J, Trouvé R et al. (2026). Pervasive increase in tree mortality across the Australian continent. Nature Plants 12, 62–73. https://doi.org/10.1038/s41477-025-02188-2
[18] Taccoen A., Piedallu C., Seynave I., Gégout-Petit A. & Gégout J. C. (2022). Climate change-induced background tree mortality is exacerbated towards the warm limits of the species ranges. Annals of Forest Science, 79(1).
[19] Lemaire J, Vennetier M, Prévosto B, Cailleret M, & Marty P. (2024). Le dépérissement du pin sylvestre dans le sud-est de la France : des interactions complexes entre climat, topographie, sol et parasite. Forêt méditerranéenne 45, 137-152.
[20] Vennetier M. (2023). Les forêts méditerranéennes face au changement climatique. Quelles leçons pour celles de la Sainte Baume ? Forêt Méditerranéenne 44, 93-108.
[21] Le Cornet L. (2025). Adaptation du massif des Maures au changement climatique. Mémoire de fin d’études, ISTOM, pp81
[22] https://www.notre-environnement.gouv.fr/actualites/breves/article/l-inrae-alerte-sur-les-consequences-du-dereglement-climatique-sur-les-feux-de
[23] Lemaire J (2025). Comprendre et modéliser le risque de dépérissement d’essences forestières dans un contexte de changement climatique. Thèse de doctorat, Aix-Marseille Université – INRAE, 351 p.
[24] Stark R & Cobb F (1969). Smog injury, root diseases and bark beetle damage in Ponderosa pine. California Agriculture, 23 (9), 13-15.
[25] Ochoa-Hueso R et al. (2017). Ecological impacts of atmospheric pollution and interactions with climate change in terrestrial ecosystems of the Mediterranean Basin: Current research and future directions. Environmental Pollution, 227, 194-206.
[26] Lemaire J, Vennetier M, Prévosto B, & Cailleret M. (2025). Multiscale modelling of european beech decline: the role of long-term climate deviations and local environmental factors. European J. Forest Research, 144, 421-441. https://doi.org/10.1007/s10342-025-01767-4.
[27] Saintonge F-X, Gillette M, Blaser S, Queloz V & Leroy Q. (2022). Situation et gestion de la crise liée aux scolytes de l’Épicéa commun fin 2021 dans l’est de la France, en Suisse et en Wallonie. Revue forestière française 73, 619-641. https://doi.org/10.20870/revforfr.2021.7201
[28] Lemaire J, Vennetier M, Prévosto B, & Cailleret M. (2022). Interactive effects of abiotic factors and biotic agents on Scots pine dieback: A multivariate modeling approach in southeast France. Forest Ecology and Management 526, 120543.
[29] Foest J, Bogdziewicz M, Pesendorfer M, Ascoli D, Cutini A, et al. (2024). Widespread breakdown in masting in European beech due to rising summer temperatures. Global Change Biology, 30, e17307.
The Encyclopedia of the Environment by the Association des Encyclopédies de l'Environnement et de l'Énergie (www.a3e.fr), contractually linked to the University of Grenoble Alpes and Grenoble INP, and sponsored by the French Academy of Sciences.
To cite this article: DIZENGREMEL Pierre, VENNETIER Michel (April 3, 2026), Forests facing global environmental change, Encyclopedia of the Environment, Accessed April 17, 2026 [online ISSN 2555-0950] url : https://www.encyclopedie-environnement.org/en/life/forests-global-environmental-change/.
The articles in the Encyclopedia of the Environment are made available under the terms of the Creative Commons BY-NC-SA license, which authorizes reproduction subject to: citing the source, not making commercial use of them, sharing identical initial conditions, reproducing at each reuse or distribution the mention of this Creative Commons BY-NC-SA license.