What is the impact of air pollutants on vegetation?
PDF
Plants are at the forefront of air pollution because they are fixed organisms at the basis of the functioning of terrestrial and aquatic ecosystems. The nature and extent of the impact of air pollutants on plants will depend on the physiological and biochemical characteristics of the affected plant, and the properties of the pollutant(s) encountered. According to the nature of the pollutant, various physiological disturbances can be observed on plants, over areas ranging from the local scale to the entire planet. They will immediately affect ecosystem functioning and in particular plant-insect relationships. They can also have effects on human health, as plants are the source of many food chains.
1. What is a polluted atmosphere?
The atmosphere, the gaseous envelope surrounding the Earth (see The Earth’s atmosphere and its gaseous envelope) contains a number of natural constituents: a mixture of gases (78% nitrogen, 21% oxygen, and also some other minor gases including 1% argon, CO2, neon, helium, ozone [1], etc.), water (solid, liquid and vapour) and solid or liquid, inorganic or organic particles in suspension (aerosols).
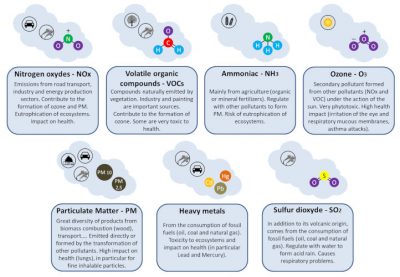
The atmosphere is polluted when the content of some of its natural constituents is higher than normal and/or when it contains new components (see Air pollution; How does the law protect air quality?; Outdoor air pollution: understanding to inform and prevent). But air pollution is mainly referred to as air pollution when these increases lead to levels of components such that they have harmful effects on the various components of different ecosystems (plants, animals, etc.), on humans (health effects) and on materials [2].
Depending on the type of component considered (Figure 1) [3], these increases may concern very small areas as well as the entire planet. If air pollution has always existed (because of volcanism, fires, for example), it is with the advent of the industrial era that it has become a real problem for the environment and health (see Air pollution; Focus Birds: flying air filters).
2. Physiological responses of plants
While plants, due to their fixed life and wide distribution, are among the first victims of air pollution, they can also be a source of secondary pollution. During high heat, they emit volatile organic compounds (VOCs) such as terpenes [4], one of the precursor gases of ozone (Figure 1). In cities in the hot regions of the USA, it is recommended not to plant certain trees (pines, oaks, etc.) in order not to increase ozone levels. Plants also emit fine particles (pollens, spores, wax compounds, various particles) which, if they have no effect on plants, can have effects on human health (allergies).
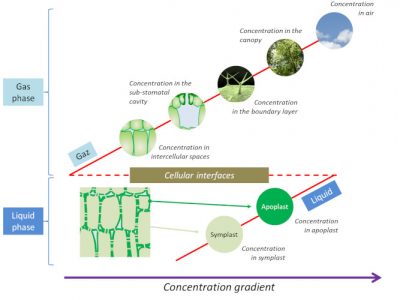
2.1. Penetration of pollutants into plants

The thickness of this layer depends on the size and shape of the leaf, the presence of leaf hairs (or trichomes) and wind speed. Its thickness is in the order of a few tenths of a millimetre.
During the temporary presence of a pollutant in this boundary layer, many reactions are likely to occur because the incident pollutant will react with:
- An aqueous phase consisting of the water film present on the surface of the leaf as well as water bound to the polar groups of the cuticle (see Focus Between protection and defence: the plant cuticles);
- A lipid phase consisting of the waxes present within (intracuticular waxes) or located on the surface (epicuticular waxes) of the cuticle (see Focus Between protection and defence: the plant cuticles);
- A gaseous phase consisting of the components of the atmosphere and the emissions of the leaf [5].
Depending on the nature of the reactions that will or will not occur at the boundary layer, the concentration of the pollutant that will enter the plant can vary greatly. Some products of these reactions are even more phytotoxic than the pollutant itself [6].
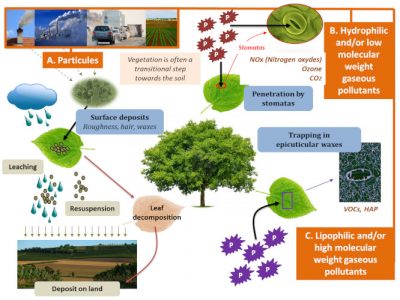
Particulate pollutants (organic or inorganic) are first captured by foliar surfaces (thanks to the micro-structure created by the presence of epicuticular waxes, trichomes, etc.), in a size range that is generally between 1 and 10 µm. In forests, this particulate deposition can vary between 280 and 1000 kg per hectare. Subsequently, meteorological conditions such as wind, sun and especially rain (leaching of leaves, dissolution of inorganic particles) influence the characteristics of this deposition (Figure 4). Thanks to the effectiveness of the cuticular barrier, organic or inorganic foliar deposition often causes only a slight penetration of pollutants into the leaves and therefore limit their physiological impacts.
After penetration, the physiological response of plants to air pollution will depend on the two actors involved: on the one hand, the characteristics of the plant and, on the other hand, the nature of the pollution.
2.2. The answer depends on the plant

Plants react to air pollution by producing reactive oxygen derivatives. After penetrating the leaves, and as for most biotic and abiotic stresses (see The fixed life of plants and its constraints; How do plants cope with alpine stresses?), the pollutants will first of all induce an oxidative stress with the production of free radicals (hydroxyl radicals) and reactive oxygen species (ROS) likely to cause damage at different levels (Figure 5) [7]. In particular, these ROS will have three main targets at the cell level: lipids (at the membrane level), proteins (at the amino acid level) and nucleic acids (adduct formation).
At the same time, the pollutant will cause specific stress related to its own physico-chemical characteristics:
- Thus, in the case of pollution by hydrofluoric acid (HF), there will be a disruption of the cellular metabolism of calcium (precipitation of calcium in the form of CaF2).
- In the case of pollution with sulfur oxides (SO2…), the reducing properties of this gas will disturb the functioning of the photosynthetic apparatus (degradation of chlorophyll)
- Acid rain, on the other hand, causes mineral deficiencies leading to yellowing of the leaves due to rainwater leaching of the mineral elements Ca, K and Mg.
When facing these stresses, the traditional strategy of defending the plant aims to limit the absorption of the pollutant and increase its tolerance to it. It consists in implementing: (a) physical processes, i.e. closing stomata, falling leaves…; and (b) chemical and biochemical processes.
These chemical and biochemical factors correspond to:
- production of insoluble precipitates (formation of CaF2 in the case of fluorine pollution);
- detoxification by the reduced form emission of the pollutant (H2S in the case of pollution by SO2, NH3 in the case of pollution by NOx);
- enzymatic degradations by cytochromes P450 and a number of antioxidant enzymes [8],[9]. Non-enzymatic antioxidant compounds such as glutathione, vitamins E and C and carotenoids may also be involved.
When a “pollution stress” is installed, the plant will therefore set up (more or less quickly) processes that will be added to the pool of defence processes already present in the plant. Following the aggression, the plant’s resistance to the pollutant will result from the combination of these various processes. For this reason, there is a specific scale of plant sensitivity for each pollutant and for each plant.
Visible and invisible damage. During low pollution and/or when the plant’s defence systems are sufficient to limit the physiological impact of a pollutant, this resistance still has a physiological cost, which is characterized by decreases in size, in yield… We then speak of “invisible damage”.
During heavy pollution and/or when the plant’s defence systems are not sufficient, irreversible damage appears such as cell death (leaf necrosis, among others). This is referred to as “visible damage” due to air pollution.
Plants react according to environmental conditions. The plant, like all biological systems, is sensitive at the same time to abiotic factors (temperature, humidity, light…) and biotic factors (age, diseases, genotypes…) in its environment. If diseases have a negative impact, other factors can have a positive impact on the plant’s response to air pollution. Thus, drought leads to the closure of stomata, which protects the plant, while the increase in CO2 promotes photosynthesis. The daily evolution of air pollution will also affect the response of plants. This is what field observations show:
- During hot weather, high temperatures lead to very high ozone concentrations in the air but at the same time to the closure of stomata. The result is a very low impact of this pollutant on vegetation during these periods. This was observed in the forests during the 2003 heat wave.
- During summer periods, ozone concentrations in the air around high altitude forests are high with slight day-night variations. As the high air humidity in these areas leads to a large opening of the stomata, a high impact of the ozone present is observed.
- On the other hand, at the level of lowland forests, air pollution is characterized by average ozone concentrations, this time with strong day-night variations. As the air humidity is lower in these areas, the opening of the stomata will be less important: for these two reasons, a lower impact of ozone is observed.
2.3. The answer depends on the pollutant

Hydrofluoric acid (HF) > ozone (O3) > sulfur dioxide (SO2) > nitrogen dioxide (NO2)
This classification is only given as an indication because there is a whole range of sensitivity of the different plants for each pollutant. For example, tobacco is very sensitive to ozone but not very affected by fluorine pollution.
In addition to the phytotoxicity of the pollutant, plants response will depend on the dose received (i.e. concentration x time). The dose is often calculated from the concentrations of pollutants in the atmosphere. It is the calculation of the flows of pollutants that have actually entered the leaf organs [10] that provides the best information on pollution-damage relationships in vegetation.
Finally, at equal doses, the shorter the application time is, the greater is the pollutant impact. This “peak effect” is usually explained by the fact that, over short periods of time, the plant does not have time to start its defence systems.
3. Symptomatology
Symptomatology analyze the signs or manifestations (symptoms) expressed by plants in response to physiological disturbances induced by air pollutants.
This symptomatology is important because it can be used as a method of bio-monitoring of air quality with plants [11]. This original method makes it possible to detect and estimate levels of air pollutants only by studying the visible (by observing necrosis) or invisible (by biochemical analyses) disturbances they cause on plants.
As the pollutants present around plants are very numerous, it is interesting to classify them, for symptomatological observations, according to the extent of their impact zone: local, regional or global.
3.1. Local pollutants
Local pollutants will have, at the plant level, impacts on at most a few tens of kilometres around their emission sources. These are mainly nitrogen compounds emitted directly from pollution sources (primary pollutants), mainly NOx (from transport) and NH3 (from agriculture and transport). These nitrogen compounds are paradoxical pollutants as they are not very phytotoxic but have a strong impact on vegetation:
- they have a beneficial fertilizing effect by promoting growth in the first place;
- but they have a negative impact over time by causing eutrophication of ecosystems [12], mineral deficiencies, effects on biodiversity (nitrophilic plants are favoured) and a decrease in resistance to various stresses.
Other local pollutants in the air are particulate deposits. It should be remembered that the vast majority of air pollution sources are both emitters of gas and dust (particles). Particulate deposits are composed of an inorganic fraction (heavy metals), but also an organic fraction (PAH, etc…). They include, among other things:
- Transport related pollutants. These are organic compounds such as BTX (VOCs in exhaust gases), or inorganic compounds such as platinoids: Pt, Rh, Pd (catalytic converters), titanium (aircraft engines), etc.
- Pollutants related to agriculture following the transfer of plant protection products such as pesticides (herbicides, fungicides, insecticides) into the air during spraying.
- Emissions from incineration facilities with in particular organic compounds such as dioxins, furans, PCBs, etc.
All these different particulate pollutants have little or no impact on vegetation, but they cause contamination of the food chains of humans and animals via plants.
For some local pollutants, the impact may be more pronounced. The massive use of more or less biodegradable detergents discharged into the sea leads to water and then air pollution (from the formation by winds of spray loaded with detergents present on the surface) [13]. The deposition of these surfactants on the leaves will then promote the penetration of salt into the plants, causing their subsequent decline and death. Impacts of this particular pollution can be observed on the edges of certain coastal forests around the Mediterranean.
3.2. Regional pollutants
Regional pollutants can have impacts over several hundred kilometres around their emission sources.They mainly include acid deposition, with mainly the presence of H2SO4 and HNO3 in wet or solid deposition. They are secondary pollutants because they are the result of interactions between primary pollutants (SO2, nitrogen compounds) and ozone [14].

Acid deposition has a low impact on plants: yellowing of needles and decrease in tree vitality due to leaching of the ions they induce. On the other hand, ozone is a very phytotoxic gas due to its direct and highly oxidizing effects on the plant’s various physiological processes (photosynthesis, respiration, etc.). It is the most worrying pollutant currently affecting vegetation and ecosystems because it causes yield losses of up to 5 to 10%, and the appearance of leaf necrosis (Figure 7) now visible in natural environments. But it also has indirect effects on vegetation because it is a greenhouse gas linked to climate change.
It is now considered that 90% of the yield losses related to air pollution in the plant world come from ozone [15]. However, this observation must be put into perspective for the growth of vegetation in anthropized ecosystems (field crops, etc.): the negative effects of ozone are very often masked by the positive effects on photosynthesis of the increase in CO2 in the atmosphere (280 ppm before the industrial era compared to more than 400 ppm currently).
Following this significant impact of ozone on vegetation and in order to detect and assess its effects, numerous lists have been drawn up indicating the sensitivity of plants present in different natural or anthropogenic environments (forests, grasslands, field crops, etc.) to ozone, depending on the climatic zones encountered (Western and Central Europe, Mediterranean coast of Europe, etc…) [16].
3.3. Global pollutants
Global pollutants have global impacts.They mainly include CO2, which is a pollutant linked to the massive use of fossil fuels by transport and industry. CO2 is a paradoxical pollutant, which will have direct beneficial effects on plant growth via its essential role in photosynthesis. But at the same time, it has indirect harmful effects on plants through the greenhouse effect and the resulting climate disturbances. Other global pollutants include:
- Methane or CH4, a gas produced by digesting herbivorous animals, anaerobic environments such as rice fields;
- CFCs and HFCs [17] (used in refrigerators or as solvents);
- N2O (from the massive use of fertilizers in agriculture);
- Methyl bromide [18] (used as a disinfectant in sericulture soils)
All these other gases only have an indirect effect on vegetation via their roles in the greenhouse effect. They are also, with the exception of CH4, ozone-depleting gases with the possibility of another negative impact on plants as a result of increased solar UV-B fluxes reaching the ground.
4. Implications for natural and human ecosystems
4.1. Impacts on ecosystems
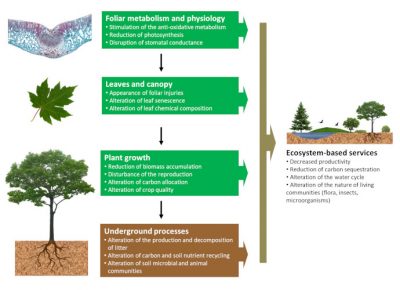
For a long time, pollution has been considered as responsible for acute toxic effects on ecosystems with sudden imbalances (massive plant mortality). Nowadays, and in our regions, the chronic effects of air pollution on ecosystems have also been observed, with progressive but often equally dangerous changes over time.
The consequences of eutrophication and the acidification of natural environments under the respective effects of nitrogen compound deposition and acid deposition are clear examples of these progressive alterations in ecosystem composition following the impact of air pollution on vegetation. Similarly, ozone -by accelerating the development and ageing of vegetation, and by reducing the vegetative cycle- will reduce the growth of plant species in natural environments with a slow repercussion on the balance of ecosystems.
Ultimately, as they move up the food chain, these slow disturbances eventually affect the entire ecosystem and lead to changes that can be very serious. We have developed (see below) the consequences resulting from this in the context of the plant-insect relationship.

4.2. Impacts on humans
The impact of air pollution on plants in natural ecosystems can have direct consequences for humans and their health, mainly as a result of the consumption of contaminated plant products from these ecosystems (fungi, berries, fruits, etc.). Indirect consequences for humans following environmental degradation are also possible: risks of erosion or landslides, possibilities of flooding and modification of the water cycle and micro-climates with the disappearance of the vegetation cover, without forgetting the negative aesthetic effects linked to biodiversity losses such as the absence of trees for example.
But it is the impacts of air pollution on plants in human ecosystems (or agrosystems), summarized in the table below, that will most directly affect humans and their health through the contamination of many food chains.
Table. Summary of the effects of pollutants on plants and their consequences for humans
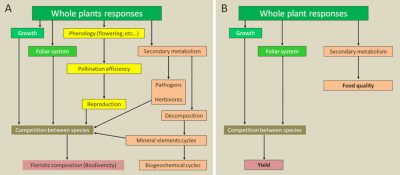
5. Impacts on plant-insect relationships
In an ecosystem, vegetation constantly interacts with its entire environment. Air pollution, by modifying plant physiology and biochemistry, will have a decisive effect on these different interactions, and in particular on plant-insect interactions. The three key plant parameters that govern these interactions – recognition, nutritional qualities and defence systems – will be affected with varying degrees of consequences.
5.1. Disturbances in the location and recognition of plants by insects

Air pollution also disrupts chemical communication between plants and insects. By acting indirectly on chemical communication substances (chemical mediators), certain pollutants such as ozone disturb plant-insect relations (e. g. recognition of spawning grounds):
- via their physiological effects on plants, for example on the emission of terpenes limited by the closure of stomata;
- by degrading or modifying the physico-chemical composition of chemical mediators, rendering them ineffective.
Finally, air pollution also causes changes in leaf recognition in insects. Pollutants such as ozone or CO2, by promoting the production of cuticular waxes and thereby modifying the physico-chemical characteristics of leaf surfaces (see Focus Between protection and defence: the plant cuticles), have repercussions on the recognition of these by insects.
5.2. Changes in plant nutritional resources for insects
Air pollution induces the presence of external elements in or on plants that can have serious consequences on associated insects. The high accumulation in plant organs of toxic air pollutants such as heavy metals, arsenic, fluorine and certain plant protection products are often the cause of poisoning and mortality of phytophagous insects (in chewing insects and “sucking insects”) and pollinating insects (bees, etc.).
In plants, air pollution – like many other stresses – causes qualitative and quantitative changes in primary and secondary metabolites. There is often an increase in concentrations of amino acids (proline), soluble proteins and sugars in the leaves, increasing their nutritional quality for some insects.
Pollutants such as SO2 and NOx, which increase sulfur and nitrogen concentrations in plants and improve their nutritional qualities, also have a positive effect on insects. This is particularly true for roadside and highway plants, which are important sources of NOx. But on the other hand, the CO2 pollution often present with NOx leads to a decrease in nitrogen concentrations in the leaves following the modification of the carbon/nitrogen ratio.
5.3. Alterations in plant defences of against insects
Phenolic compounds and foliar cuticle, which constitute respectively a chemical defence system and a physical defence barrier for plants, are likely to be modified by pollutants:
- Ozone and CO2, like many other air pollutants that induce oxidative stress, increase concentrations of phenolic compounds in plants, thereby reducing the nutritional and taste qualities of leaves for insects.
- At the same time, by promoting the production of cuticular waxes, both ozone and CO2 improve the characteristics of the physical barrier represented by the cuticle (see Focus Between protection and defence: the plant cuticles).

6. Messages to remember
- Gaseous air pollutants enter plants through leaf stomata while particulate pollutants are captured by the micro-structure of leaf surfaces.
- The phytotoxicity of air pollutants depends on their chemical nature.
- The pollutants induce oxidative stress in the plant with the production of free radicals (hydroxyl radicals) and reactive oxygen derivatives (ROD) that cause damage in the leaf.
- The plant uses processes such as stomatal closure to limit the absorption of the pollutant.
- Some external factors can have a positive impact on the plant’s response to air pollution. Thus, drought leads to the closure of stomata, which protects the plant.
- Air pollution, by modifying plant physiology and biochemistry, has a decisive effect on ecosystems, including plant-insect interactions.
- Nitrogen compounds are not very phytotoxic but have a strong impact on vegetation: a beneficial fertilizing effect in the first place and a negative impact in the long term by causing eutrophication of ecosystems.
- 90% of the yield losses due to air pollution in the plant world come from ozone.
- While the various particulate pollutants have little or no impact on vegetation, they contaminate the food chains of humans and animals through the plants that are consumed.
- Air pollution alters the nutritional qualities of plants used by plant insects.
- Plant defences against insects are altered by air pollution.
References and notes
Cover image. Tobacco leaf necrotic under the action of ozone. [Source: © J.P. Garrec]
[1] In the case of air pollution, we refer to ground-level ozone, a secondary pollutant formed in the lower layers of the atmosphere, near the earth’s surface. This ozone is to be distinguished from ozone found at a fairly high concentration in the Earth’s stratosphere, mainly at an altitude of between 15 and 20 km. By absorbing nearly 97% of the ultraviolet rays from the sun, stratospheric ozone forms a layer that protects living organisms from the dangers of UV radiation.
[2] https://www.airparif.asso.fr/pollution/effets-de-la-pollution-batiment
[3] It should be noted that NOx and NH3 produce PM by direct gas-solid conversion, by nucleation with water drops, and also by reaction of NH3 on NOx to form ammonium nitrate.
[4] Misztal, P.K., Hewitt, C.N., Wildt, J., Blande, J.D., Eller, A.S.D., Fares, S., … Goldstein, A.H. (2015). Atmospheric benzenoid emissions from rival plants those from fossil fuels. Scientific Reports, 5, 12064. http://doi.org/10.1038/srep12064
[5] These are on the one hand natural emissions (H2O, CO2, terpenes, isoprenes), on the other hand stress related emissions (ethylene) and finally emissions from the biological reduction of pollutants: H2S formed from SO2, NH3 formed from NOx.
[6] This is the case of the reaction between ozone and components of epicuticular waxes (unsaturated hydrocarbons) that produce ozonides and hydroxyhydroperoxides (HHP), and the O3 +C2H4 reaction that leads to the formation of hydroxymethyl hydroperoxide (HMHP).
[7] Baier M., Kandlbinder A. Golldack D. & Dietz K.J. (2005) Oxidative stress and ozone: perception, signalling and response. Plant, Cell & Environment, 28(8), 1012-1020.
[8] For example, superoxide dismutase enzymes, catalases or ascorbate peroxidase.
[9] Foyer CH, Noctor G. (2005) Redox homeostasis and antioxidant signaling: a metabolic interface between stress perception and physiological responses. Plant Cell. 17, 1866-1875.
[10] Taking into account the diffusion resistance of the boundary layer and stomata.
[11] Garrec J.P. & Van Haluwyn C. (2002) Biosurveillance végétale de la qualité de l’air. Concepts, méthodes et applications. Editions Tec et Doc Lavoisier, Paris, 118 pages.
[12] Eutrophication caused by atmospheric nitrogen deposition is currently the most significant impact of air pollution on ecosystems and biodiversity. Eutrophication of terrestrial ecosystems due to air pollution. Annual Indicator Report Series (AIRS), In support to the monitoring of the 7th Environment Action Programme. As of November 30, 2017; Jones L., et al. (2014) A review and application of the evidence for nitrogen impacts on ecosystem services. Ecosystem Services, 7, 76-88, ISSN 2212-0416.
[13] Garrec J.P., Sigoillot I.E. (1992) Les arbres malades de la mer. La Recherche, 245, 940-941.
[14] Ozone is also a secondary pollutant resulting from complex reactions under the action of the sun between different gaseous pollutants emitted – in particular – by transport: VOCs, NOx.
[15] Holland M., Kinghorn S., Emberson L., Cinderby S., Ashmore M., Mills G., Harmens H. (2006) Ozone and Crop Losses 2006 (ICP Vegetation Report for Defra Contract EPG 1/3/205).
[16] See ICP vegetation reports; Mills G., Hayes F., Jones M.L.M. & Cinderby S. (2007). Identifying ozone-sensitive communities of (semi-) natural vegetation suitable for mapping exceedance of critical levels. Environmental Pollution 146: 736-743.
[17] In 1987, in Montreal, the main CFC-producing countries decided to stop producing CFCs. In Europe, CFCs have not been available on the market since 2000 and have had to be recovered and destroyed since 2002. To replace CFCs, manufacturers then adopted HFCs, which have been widely used but also have an environmental impact. The end of the use of HFCs was recorded in an agreement signed in Kigali (Rwanda) in 2016.
[18] Methyl bromide is one of the substances covered by the Montreal Protocol (1987). The production and placing on the market of methyl bromide has been banned since 2005, except for derogations for strictly regulated uses.
The Encyclopedia of the Environment by the Association des Encyclopédies de l'Environnement et de l'Énergie (www.a3e.fr), contractually linked to the University of Grenoble Alpes and Grenoble INP, and sponsored by the French Academy of Sciences.
To cite this article: GARREC† Jean-Pierre (January 5, 2025), What is the impact of air pollutants on vegetation?, Encyclopedia of the Environment, Accessed June 6, 2026 [online ISSN 2555-0950] url : https://www.encyclopedie-environnement.org/en/life/impact-air-pollutants-on-vegetation/.
The articles in the Encyclopedia of the Environment are made available under the terms of the Creative Commons BY-NC-SA license, which authorizes reproduction subject to: citing the source, not making commercial use of them, sharing identical initial conditions, reproducing at each reuse or distribution the mention of this Creative Commons BY-NC-SA license.