Cellular impact of solar UV rays
PDF
The Sun is essential to life, whether through photosynthesis in plants or the production of vitamin D in humans. However, it is a source of harmful effects linked particularly to the ultraviolet (UV) portion of its electromagnetic emission spectrum. Indeed, solar UV induces damage to cellular components and in particular to DNA. In plants and microorganisms, the consequences are mainly observed in terms of growth. In humans, the major consequences are mainly the induction of skin cancers and skin ageing, as well as eye damage.
1. The Sun, a complex source of radiation
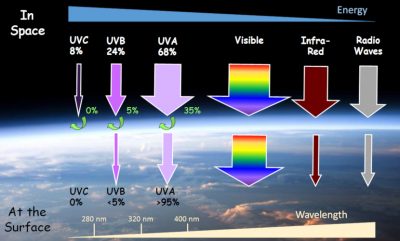
The electromagnetic radiation present in the solar spectrum (Read Solar energies) before passing through the Earth’s atmosphere extends over a wide wavelength range (Figure 1):
- Radio waves;
- Infrared radiation, responsible for the sensation of heat;
- Visible light;
- The ultraviolet (UV) portion.
The latter is arbitrarily decomposed into several zones according to the energy of the photons, which is measured by the wavelength, a physical parameter inversely proportional to the energy of the radiation. UVC photons exhibit a wavelength of less than 280 nm, UVB between 280 and 320 nm, and UVA between 320 and 400 nm. Outside the atmosphere, the respective proportions of these different UV radiations are 8, 24 and 68%. This distribution changes significantly after crossing the atmosphere, particularly because of the ozone layer, which absorbs all UVC (the most energetic UV rays), a large part of UVB and a fraction of UVA. On the Earth’s surface, the proportions of UVC, UVB and UVA are thus 0, 5 and 95%. This ratio between UVB and UVA also depends on other factors such as latitude (more UVB at the equator), altitude (more UVB in the mountains), time of day (more UVA in the morning and evening) and albedo (UV reflection on the ground). Sunlight is therefore to a source of various UV radiations, all classified as carcinogenic by the World Health Organization (WHO), to which we are constantly exposed.
2. DNA, an essential and fragile cellular component
One of the major deleterious effects of overexposure to the sun is the induction of skin cancers. The initial event in the formation of a skin tumor is often DNA damage. DNA plays a major role in the life of the cell. First of all, it is “the recipe book” for making the proteins that provide the structure and various cellular functions. Copied from mother cell to daughter cell, it also ensures the transmission of genetic information. These essential functions are ensured by its chemical structure: two strands made up of a phosphate and sugar skeleton on which small cyclic molecules are grafted, the four bases of DNA (adenine A, thymine T, guanine G and cytosine C). It is the order in which the latter are linked inside the genes that constitutes the support of heredity. Yet this beautiful machinery is permanently endangered by various chemical or physical agents that have the ability to modify the structure of DNA. Indeed, the enzymes responsible for translating or replicating DNA block or make mistakes as soon as they encounter DNA damage or lesions (see Genetic polymorphism and variation). Unfortunately, UV radiation from the sun is one of those agents that is particularly effective in damaging our DNA and threatening the integrity of our genome.
3. UVB, the least frequent but most aggressive
How can a single photon damage a structure such as the DNA double helix? The underlying mechanisms actually depend on the energy of the incident radiation. The damage induced in DNA by UVB radiation is well known and its formation is relatively simple. Indeed, the DNA bases efficiently absorb UVB photons and find themselves with an excess of energy, in a physical state logically qualified “excited”. One of the solutions to return to their fundamental state is to react chemically with the neighbourhood.
In this case, it is the bases of the pyrimidine class, thymine (T) and cytosine (C) that are the most reactive. Thus, an excited pyrimidine can create covalent bonds with a neighbouring pyrimidine. Dimeric photoproducts (because they are induced by light and involve two bases) of pyrimidines (two T, T and C, or two C) are then created. The major role of UVB and the photoproducts it induces in DNA is now well established. Indeed, there are produced in large quantities (biologically speaking), in the order of 1 dimer per million bases, in the DNA of skin exposed to the sun.
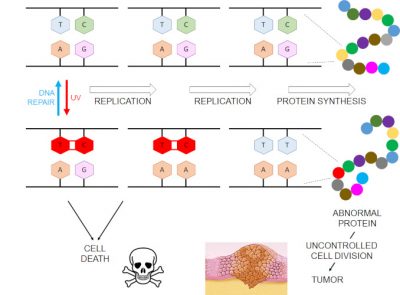
To defend themselves against these changes in the chemical structure of DNA, cells have repair systems. In humans, the mechanism for the elimination of pyrimidine dimers is based on the excision of a small DNA fragment containing the damage and resynthesis of “new” DNA using the undamaged strand as a template. A deficiency in this repair system is highly predisposing to skin cancers, for example in the case of the genetic syndrome Xeroderma pigmentosum. DNA repair, even in normal cells, is not perfect, however, and cell divisions can occur in the presence of DNA damage. The enzymes responsible for replicating the latter have evolved to use natural ATGC bases and may mistranslate the damaged portion of DNA, thus inducing a modification of the gene sequence (Figure 2). It is a mutation that will be passed on to subsequent generations of cells. If this mutation occurs in a gene that controls cell division, a tumor process may be triggered. Thus, it is at DNA sites with two adjacent pyrimidine bases that mutations in skin tumours are found, indicating the major role played by dimeric photoproducts. DNA photoproducts can also induce cell death, sometimes voluntarily as in the case of apoptosis. This appears to be a lesser evil, as the cells that are too damaged are removed from the tissue.
4. UVA rays, not so harmless radiation
Long neglected or considered mainly for its involvement in skin aging, UVA is now recognized as being capable of inducing mutations and tumors. Although its effectiveness is less than that of UVB, UVA rays are at least 20 times more abundant. The major effect of UVA in skin cells is the production of oxidizing chemical species, such as the highly mediatized “free radicals” or the most discreet but equally effective “singlet oxygen“, an activated form of molecular oxygen. These compounds are produced by photosensitization processes that are triggered when certain cellular components absorb UVA and transfer the absorbed energy to oxygen or remove electrons from nearby molecules.
The reactive oxygen species thus generated can then damage the cellular macromolecules: lipids, proteins… and DNA. The nature of DNA oxidation products is very varied. Some free radicals can damage the sugars in the biopolymer and induce breaks. Free radicals and singlet oxygen can also modify the bases. Guanine is in particular a weak link because of its strong ability to get oxidized. More complex damage involving the formation of covalent bonds between DNA and proteins is also produced. The important role of this oxidative stress has led to the development of photoprotection strategies based on the ingestion of antioxidants.
The reality of the effects of UVA is more complex than the mere induction of oxidative stress as it is often highlighted. Indeed, it has been shown in recent years that UVA can also induce pyrimidine dimers in DNA. Although the yield is much lower than with UVB, dimers are still more frequent than oxidation products in the DNA of cells exposed to UVA. Finally, it has been reported that in human cells, UVA, like UVB, induces mutations mainly at sites containing two pyrimidines. Although UVA is less harmful than UVB, its contribution to the genotoxic properties of sunlight cannot be neglected. In particular, these data must be taken into account when considering a reduction in the use of artificial tanning lamps, which are very rich in UVA. Their use can be considered as a voluntary exposure to a mutagenic and carcinogenic agent with no health benefit.
5. The need for effective photoprotection
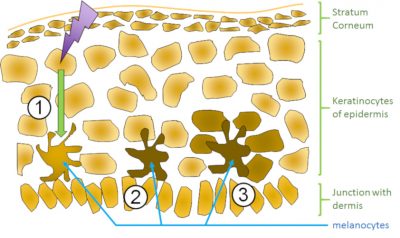
It therefore appears that solar UV radiation has harmful properties for health. It is of course inconceivable to imagine a drastic limitation of exposure to the Sun that is part of our daily environment. In addition, a reasonable dose of UV rays is necessary, for example, for the production of vitamin D, which is largely the result of a photochemical reaction in the skin. However, it is essential to avoid overexposure and develop photoprotection strategies. A first obvious possibility is the natural pigmentation reaction of the skin induced by sunlight and in particular UVB, i.e. tanning. The latter results from the production of melanin, a polymer over absorbing light over a wide spectrum from UV to visible. DNA damage plays a role in this mechanism. Indeed, it is following the formation of pyrimidine dimer that keratinocytes, the cells present in the outer layer of the skin (the epidermis) send a signal to melanocytes, the cells where melanin is synthesized (Figure 3). Melanin is then transported via dendrites to the keratinocytes, thus providing a photoprotector within the tissue itself. However, tanning does not guarantee absolute protection. First of all, it only corresponds to a sunscreen with an index of 3 to 5. In addition, melanin production is a response to aggression and DNA damage. Some dermatologists even go so far as to talk about scars for tanning. In short, if tanning is a natural damage-limiting reaction, it is not a total protection. It should be noted that the tan obtained in tanning salons is even less satisfactory. More than the production of new melanin, the colour that the skin takes is due to the oxidation by UVA of the pre-existing melanin.
The use of other means is therefore necessary to avoid the health impact of excessive exposure. The first is simple in principle and consists in adopting reasonable behaviour by avoiding overexposure. Unfortunately, this is thwarted by the current aesthetic canons and recreational practices that push a part of the population to expose themselves to the periods with the highest UV rays. In addition, a large number of professions require very frequent exposure to the sun (farmers, construction workers, etc.). In both cases, it is necessary to reduce the amount of UV rays reaching the skin. The most effective practice is the use of appropriate clothing and hats. Again, these practices are not always easy to get accepted and the use of sunscreens is becoming interesting. Recent commercial products have incorporated enhanced protection against UVA rays, while older screens were mainly directed against UVB rays. Many studies showed their effectiveness both against sunburn, a phenomenon used to determine their protection index (SPF), but also against the formation of DNA damage. However, these works are carried out under standardised conditions with the application of large quantities of product. In real life, the quantities used are much smaller and the protection factor can be 3 or 4 times lower than that determined by the manufacturer. The use of high-index products is therefore probably necessary. Despite this, it should be kept in mind that no formula totally absorbs UV rays (there is no longer any total sunscreen!) and that sunscreens should be considered as a way to limit the consequences of exposure but in no way as a pretext to prolong it.
6. Facing the most common carcinogenic agent: solar UV radiation
Solar ultraviolet radiation is probably the most common mutagenic and carcinogenic agent to which humans are exposed. Although the chemical and biological mechanisms involved in this toxicity have been studied for a long time, new explanations continue to be highlighted. It should be noted that other components of the environment such as plants and microorganisms are also affected. The strategies developed by these organisms to defend themselves are sometimes different from those used by mammals. Particular emphasis should be placed on plants that need sunlight to grow but must limit the associated damage. Thus, a large part of their defence against DNA damage is based on light-activated repair systems, thus using the mutagenic agent to counteract its harmful consequences. All this shows the strong impact of solar UV on the environment, and therefore on mankind.
References and notes
Cover image. Wheat field behind St. Paul’s Hospice with a mower and sun, Vincent Van Gogh, 1889; Folkwang Museum, Essen, Germany
The Encyclopedia of the Environment by the Association des Encyclopédies de l'Environnement et de l'Énergie (www.a3e.fr), contractually linked to the University of Grenoble Alpes and Grenoble INP, and sponsored by the French Academy of Sciences.
To cite this article: DOUKI Thierry (January 5, 2025), Cellular impact of solar UV rays, Encyclopedia of the Environment, Accessed May 29, 2026 [online ISSN 2555-0950] url : https://www.encyclopedie-environnement.org/en/life/cellular-impact-of-solar-uv-rays-2/.
The articles in the Encyclopedia of the Environment are made available under the terms of the Creative Commons BY-NC-SA license, which authorizes reproduction subject to: citing the source, not making commercial use of them, sharing identical initial conditions, reproducing at each reuse or distribution the mention of this Creative Commons BY-NC-SA license.







