The path of carbon in photosynthesis
PDF
How is the carbon from carbon dioxide – CO2 – present in the atmosphere integrated into the organic matter that makes up living organisms, the biomass? For several billion years, this process has been carried out during the biochemical stages of photosynthesis by organisms using the energy recovered from sunlight by chlorophyll. However, photosynthesis has had to adapt these mechanisms to survive the various environmental changes that have taken place over geological time scales. The accumulation -in the atmosphere- of oxygen (O2) produced during the photochemical stages of photosynthesis (see Shedding light on Photosynthesis) was one of these major events. Different original strategies have been adopted over the course of evolution, and have thus made it possible to produce an immense biodiversity of organic biomolecules that we use for food, heating, clothing, housing and health care.
1. What is photosynthesis?
1.1. Making biomass from CO2 in the air
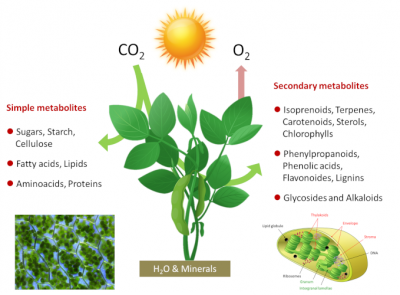
Photosynthetic organisms are therefore photoautotrophic*. Photosynthesis is at the origin of most of the molecules in the food chain of living beings and the majority of the organic biomass of our Planet. Photosynthesis takes place in chloroplasts, green intracellular organelles a few micrometers in size that contain the photosynthetic machinery (see Shedding light on Photosynthesis). The simplified equation for photosynthesis can be written as follows:
CO2 + H2O + light energy → energy-rich carbon molecules + O2
Photosynthesis fixes 115 to 120 billion tons (or Gigatons) of carbon each year from CO2 in the air, including 60 for the continents. To achieve this, plants use a very small part (about 1-2%) of the solar energy reaching our planet. On a global scale a power of about 130-140 terawatts (1 terawatt = 1012 watts) is used, which is about six times the energy consumption of mankind. How do photosynthetic organisms achieve this?
1.2. Photosynthesis is divided into two phases
- a very fast photochemical phase that takes place in the membrane system of the chloroplasts, the thylakoids (Figures 1 & 2).
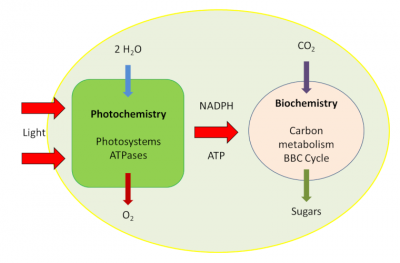
- A metabolic phase, slower than the previous one, takes place in the inner liquid of the chloroplasts, the stroma (Figure 2).
The biochemical mechanisms involved in the fixation of carbon from the CO2 of the air require the presence of a carbon receptor: an enzyme that ensures this fixation and carboxylation, giving rise to organic compounds. This photosynthetic carbon metabolic pathway is known as the Benson-Bassham-Calvin Cycle.
This article focuses primarily on the description :
- the biochemical mechanisms of photosynthesis responsible for the fixation of carbon from carbon dioxide in the atmosphere ;
- of their evolution during changes in the environment;
- the impact of the appearance of oxygen in the atmosphere during different geological periods.
2. How do plants fix carbon from CO2?
2.1. Some history
Jean Sénebier (see Focus Some pioneers in photosynthesis) was the first scientist having stated -as early as 1782- “that carbon dioxide CO2 is fixed under illumination by photosynthetic organisms and represents food for the plant“. From the late 18th century to the mid-1940s, the nature of the photosynthetic pathways for the assimilation of carbon from carbon dioxide (CO2) remained a mystery. It was first assumed -in the early 19th century- by J.B. Boussingault and F. Bayer, that carbohydrates could result from the combination of carbon with the elements of water, hence the first name carbohydrates was given to sugars. Most formulas representing sugars can in fact be inscribed as if they were the result of the polymerization of this fundamental molecule containing carbon and water: (CH2O)n. Subsequently, several other compounds were mentioned as the first products of photosynthesis. Examples include carbonic acid H2CO3, formic acid HCOOH, the simplest of the carboxylic acids*, etc. However, there were no experimental results to confirm these hypotheses. If the hypothesis of formic acid has been maintained for a very long time in the literature as the first product of photosynthesis, it owes this only to its disarming simplicity.
2.2. The Benson-Bassham-Calvin Cycle
- Search for CO2 acceptor and discovery of a specific carboxylase
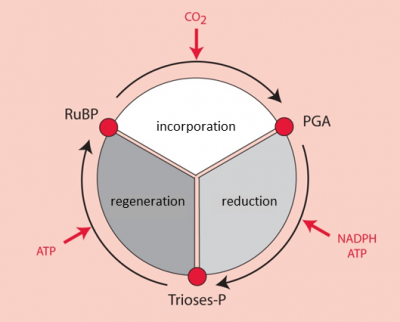
It was by using 14CO2 as a radioactive tracer that Benson [2] observed that the carbon of 14CO2 binds to a more complex pre-existing carbon structure: a five-carbon phosphoryl compound, ribulose-1,5-bisphosphate or RuBP (see Focus on Deciphering the Benson-Bassham-Calvin Cycle). [3] This compound has a chemical structure favourable to the addition of a carbon (a reaction called carboxylation). This reaction results in the formation of a very unstable six-carbon compound, which is immediately metabolized into two three-carbon (C3) molecules, phosphoglyceric acid, PGA [4] (Figure 3).
The enzyme that binds the CO2 carbon to RuBP is a carboxylase*, RuBP carboxylase, later called RubisCO (see below). This specific enzyme of photosynthesis has been the gateway for carbon to enter the majority of the planet’s organic molecules for more than three billion years (see Focus RubisCO).
RubisCO, a complex enzyme of high molecular weight (550 kDa) [5], is localized in the stroma of chloroplasts where it accounts for 30-50% of soluble proteins. RubisCO is the most quantitatively important enzyme in the biosphere, and is thus the main reserve of organic nitrogen in leaves [6]. Because of its central role in autotrophy, it is considered that the presence on Earth of each human being required the formation of 5 kg of RubisCO.
- Reduction of phosphoglyceric acid to triose-phosphates
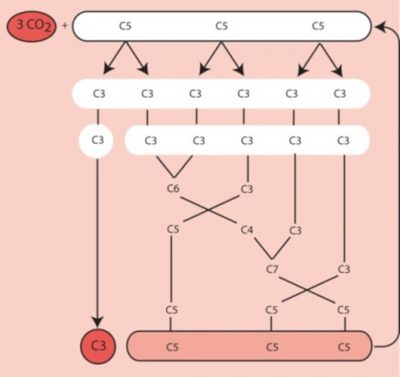
- Fate of triose-phosphate
For six molecules of triose-phosphate synthesized, only one is intended for the synthesis of carbohydrates, amino acids, lipids, etc. The other five molecules of trioses-phosphate are used to regenerate RuBP, the CO2 acceptor (Figure 4). The regeneration of a RuBP molecule has a high energy cost that requires 2 NADPH and 3 ATP per molecule but this energy is provided free of charge by the Sun.
Since the first products of this ring are three-carbon molecules, the plants using this ring have been called C3-type photosynthetic plants.
Trioses-phosphate that are not used for RuBP recycling are either (a) used in the chloroplast for the synthesis of starch, amino acids or lipids or (b) exported out of the chloroplast and transformed into sugars by the enzymes of the cytoplasm for further metabolism (see Focus Sucrose or Starch?).
- Synthesis and transport of photosynthetic assimilates
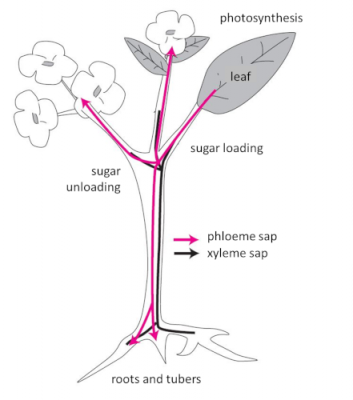
2.3. What about temperature?
Temperature affects biophysical and metabolic processes differently. Biophysical processes such as light absorption by chlorophyll pigments and the formation of NADPH and ATP are not very sensitive to temperature changes. On the other hand, the biochemical reactions that cause CO2 and O2 fixation and sugar synthesis, as well as the exchange of molecules between cell compartments and organs, are highly dependent upon it. On average, a 10°C rise in temperature doubles the velocity of biochemical reactions.
In temperate regions, air and plant temperatures are subject to strong seasonal and daily variations that are parallel to the amount of solar energy reaching the ground surface. Plants can, to varying degrees, adapt to rapid daily variations in temperature, between morning and end of day for example. Rapid changes in leaf temperature usually follow variations in sunlight. In environments characterized by low temperatures, such as the alpine environment, plants have developed mechanisms that allow them to cope with these temperature variations (see How do plants cope with alpine stresses? ).
Plants can also acclimatize to long-term temperature changes. In all cases, the temperature at which maximum photosynthetic activity is observed follows the growth temperature. Acclimatization to new thermal conditions can nevertheless cause a decrease in photosynthesis in some plants.
3. Oxygen production over geological times
The first photosynthetic reactions appeared more than three billion years ago when the atmosphere was almost devoid of dioxygen O2 but composed mainly of water (H2O), carbon dioxide CO2 (10 to 15%), nitrogen dioxide (N2), and hydrogen sulfide (H2S). At that time, during the transformation of light energy into energy-containing molecules, primitive photosynthetic bacteria – the purple sulphurous bacteria like the green sulphurous bacteria – oxidized hydrogen sulphide. Photosynthesis was of an anoxygenic type.
With the appearance of the ancestors of cyanobacteria, H2O became the almost inexhaustible substrate for oxidation and the supplier of electrons and protons leading to oxygen release into the atmosphere. Photosynthesis became of the oxygenic type (see Shedding light on Photosynthesis). Presently, these two types of photosynthesis coexist:
CO2 + 2 H2S → (CH2O) + H2O + 2 S (Anoxygen photosynthesis)
CO2 + 2 H2O → (CH2O) + H2O + O2 (Oxygenic photosynthesis)
After the onset of oxygen-source photosynthesis about 2.5 billion years ago, the concentration of O2 in the atmosphere remained very low for a very long period of time due to the high capacity of crust minerals to trap oxygen in the form of iron oxide (Fe2O3). This phase in the Earth’s history is clearly marked in the red geological layers rich in this iron compound (see The Biosphere, a major geological player). After all the minerals were saturated by oxygen, i.e. after the “great oxidation” period that took place about 2.4 billion years ago, the level of oxygen released by the photosynthetic activity of cyanobacteria and eukaryotes strongly increased in the atmosphere. At concentrations close to 21% of the gaseous concentration in the atmosphere, the oxygen content has become a serious issue for photosynthetic species.
4. Oxygen, a catastrophe for photosynthesis?
4.1. More history
In the 1920s, Otto Warburg [7] observed that if the oxygen O2 content of the air (currently 0.0408% CO2) [8] is lowered by 20 to 2%, the net rate of CO2 assimilation is multiplied by a factor of 1.5 to 2. This is the so-called Warburg effect: high oxygen tensions inhibit carbon dioxyde uptake under illumination. In the 1970s, following labelling experiments using the oxygen isotope 18O, Bowes, Lorimer, Ogren and Tolbert showed that ribulose biphosphate carboxylase, the enzyme that binds carbon dioxide, is also capable of binding oxygen. [9]
4.2. Dilemma for the RubisCO: the O2/CO2 competition
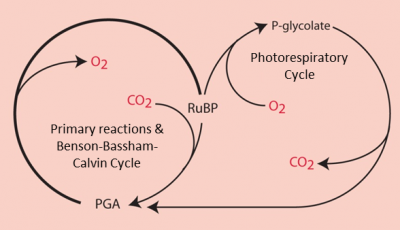
- Carbon dioxide promotes the carboxylase function of the RubisCO ;
- Dioxygen promotes the oxygenase function through a process called photorespiration.
In the presence of a high concentration of CO2, RubisCO functions only as a carboxylase leading to the synthesis of two PGA molecules (C3 molecules), which are the origin of the phosphorylated sugars formed by the Benson-Bassham-Calvin ring.
RuBP (C5 molecule) + CO2 (C1 molecule) → 2 PGA (C3 molecule) (carboxylation reaction)
On the other hand, in the presence of a high concentration of O2 and a low concentration of CO2, RubisCO gives rise to a PGA molecule (C3 molecule) and a molecule with two carbon atoms, the phosphoglycolate (or P-glycolate).
RuBP (C5 molecule) + O2 → PGA (C3 molecule) + P-glycolate (C2 molecule) (Oxygenation reaction)
4.3. The 2P-glycolate Cycle
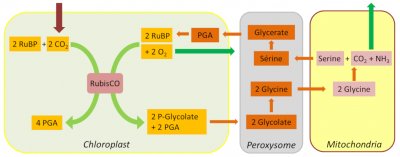
In addition to these carbon and nitrogen losses, glycolate recycling also has a significant energy cost in NADPH and ATP. However, thanks to the unfolding of the glycolate pathway, a large part of the carbon from photorespiration is eventually recovered, thus limiting the loss of photosynthetic carbon (Figure 7).
Photorespiration is mainly expressed in plants growing in temperate regions (wheat, barley, tomato, lettuce, potato, trees), photosynthetic plants of type C3. It is estimated that at 25°C, under normal environmental conditions, i.e. 21% oxygen and 0.0408% CO2, the ratio between carboxylation and oxygenation rates is about 2.5, i.e. the emission of photorespiratory CO2 corresponds to about a 20% loss of photosynthetic CO2 assimilation. The importance of photorespiration is very much linked to environmental conditions:
- Photorespiration is all the more important as the temperature and the illumination are high and the CO2 content of the atmosphere is low;
- Conversely, high CO2 concentrations favour carboxylation.
4.4. Photorespiration: a major adaptive process
For more than 3 billion years, photosynthesis, a very robust process, has been very stable while adapting to the major environmental changes that the planet has undergone (see The Biosphere, a major geological player). The evolution of photosynthetic metabolism is tightly associated with changes in the environment:
- As the oxygen content of the atmosphere increased, the CO2/O2 ratio decreased dramatically and caused global glaciation about 700 million years ago.
- These new conditions induced a high oxygen pressure on the functioning of RubisCO in microorganisms and algae, prior to the colonization of the continents. [11]
- Adapting to these new conditions, the branch of the green lineage (ancestor of terrestrial plants) developed the photorespiratory pathway, which in turn enabled the subsequent colonization of the continents, some 430 million years ago.
- Once in the open air, plants have had to cope with this new evolutionary pressure and have sought to reduce or bypass photorespiration by different strategies.
Photorespiration is thus an inevitable photosynthetic process because it is linked to the intrinsic properties of the RubisCO itself that formed during evolution at a time when the oxygen content of the environment was almost negligible [12].
5. Concentrating CO2 in the vicinity of the RubisCO
Apart from C3 plants, several other photosynthetic organisms (cyanobacteria, C4 and CAM plants…) have developed original strategies to effectively reduce the harmful effects of oxygen on RubisCO. One of the most ovious was to concentrate CO2 close to the enzyme.
5.1. Photosynthetic bacteria: creating a CO2 reservoir close to the RubisCO
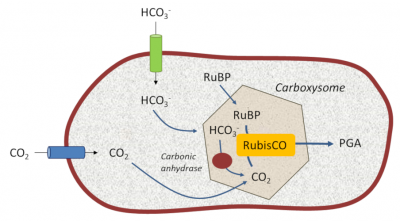
These structures allow cyanobacteria to live in aquatic environments that are poor in dissolved CO2 but rich in bicarbonate HCO3– ions. Specific and efficient transporters, located on their limiting membrane, capture bicarbonate (HCO3–) which they transform into CO2 thanks to specific enzymes called carbonic anhydrases*. This mechanism creates an internal reservoir of concentrated carbon dioxide in the environment close to their RubisCO, thus somehow recreating the primitive atmosphere of ancient geological times. This promotes the carboxylase activity of RubisCO at the expense of the oxygenase activity. [13]
5.2. How to physically separating CO2 fixation and RubisCO? the solution of C4 plants
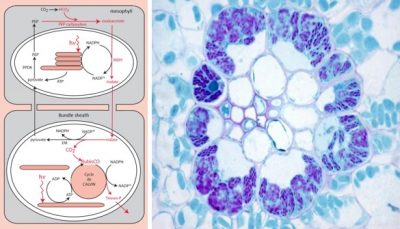
- One surrounding the conducting vessels, the outermost tissue, the mesophyll;
- The other one surrounding the most internal tissue, the perivascular sheath (a very impervious russian nesting dolls type of structure).
Mesophyll cells contain specific carboxylases, phosphoenol-pyruvate-carboxylases or PEP-carboxylases, which catalyze the formation of a four-carbon compound (hence the name photosynthesis or C4-type plants), oxaloacetate [14] :
PEP (C3 molecule) + Bicarbonate (C1 molecule) → Oxaloacetate (C4 molecule)
In the mesophyll chloroplast, oxaloacetate is transformed into another C4 compound, malate, and migrates into the cells of the sheath. There, after enzymatic decarboxylation of this four-carbon compound, a significant amount of CO2 accumulates in the environment close to the RubisCO, promoting its carboxylase activity. The phosphoenol-pyruvate is then regenerated to ensure the durability of the cycle.
This mechanism – which physically separates the capture of atmospheric carbon dioxide and its use by the RubisCO – has, however, an additional energy cost in ATP compared to the C3 mechanism of photosynthesis. [15]
5.3. Temporal separation in succulent plants: C4 metabolism at night and C3 during the day
In succulent plants (cacti, pineapples, etc.) or more generally plants with the CAM-type (Crassulacean acid) metabolism, the functions of CO2 concentration and carboxylation of RubisCO are located in the same tissue. But there is a temporal separation of their functioning: at night C4 metabolism is active, ensuring malate synthesis, whereas during the day C3 metabolism is active owing to CO2 released by malate decarboxylation. [16] (see Focus Joubarbe: example of a plant’s adaptation to environmental constraints).
6. Photosynthesis in a changing environment
6.1. How metabolic types favor plants adaptation to environmental changes?

Under high illumination and high temperatures, C4 plants -showing virtually no photorespiratory activity- are more efficient at assimilating carbon from atmospheric CO2 than C3 plants, provided that water and minerals are not limiting. For example, the 5% of C4 plant species on the planet fix 30% of the world’s CO2. And furthermore:
- For the same biomass production, C4 plants use at least one third less water due to their sleeve leaf structure. Only 350 litres of water are needed to produce 1 kg of maize (a C4 plant, Figure 10) flour compared to 500 litres of water for 1 kg of wheat (a C3 plant, Figure 11) flour;
- C4 plants mobilize less nitrogen than C3 plants because the efficiency of PEP-carboxylases allows to reduce the quantity of RubisCO -an enzyme very rich in nitrogen-, to reach the same photosynthetic activity as C3 plants.

Moreover, if the concentration of CO2 continues to rise in the atmosphere as it is currently observed (see A carbon cycle disrupted by human activities), C3 plants are expected to reach photosynthetic activities approaching those of C4 plants provided that temperatures remain moderate.
6.2. And in the future?
This observations suggests that, over the coming decades, plants will most likely acquire mechanisms for adaption to their changing environment.
Our better knowledge of the different mechanisms by which plants adapt to environmental changes allows us to consider developing plants that could be better adapted to rapid changes in CO2 content, temperature rise, water availability, etc. Of the many research projects currently underway, it is not yet known which of them will prove to be profitable and suitable for large-scale agricultural or industrial application. The focus Improving photosynthesis? presents some possible directions.
7. Messages to remember
- Through photosynthesis, plants and certain bacteria convert part of the sunlight into stable chemical energy and simultaneously fix the carbon dioxide CO2, so as to elaborate all the organic molecules necessary for the development of life.
- The use of radioactive 14C as a molecular marker and the development of analytical techniques have made it possible to deciphering the carbon metabolic pathway and to highlight the Benson-Bassham-Calvin Cycle. This cycle ensures the regeneration of the carbon acceptor of CO2 and the synthesis of the elementary molecules at the origin of sugars, proteins and lipids necessary for the elaboration and functioning of photosynthetic cells.
- The carbon fixation of CO2 which integrates the Benson-Bassham-Calvin Cycle has been catalyzed for several billion years by a specific enzyme of photosynthesis, ribulose bisphosphate carboxylase (RuBP carboxylase).
- As a result of the increase in the planet’s oxygen content (in the atmosphere and the oceans), the RuBP carboxylase has also fixed oxygen, thus manifesting not only a carboxylase function but also an oxygenase function, hence its name RubisCO.
- The oxygenase function is responsible for the synthesis of a phospho-glycolate molecule, a powerful inhibitor of the Benson-Bassham-Calvin Cycle. Plants in the course of evolution have retained a metabolic pathway that eliminated 2P-glycolate with CO2 emission, hence the name photorespiratory cycle.
- Other photosynthetic organisms have developed more original and effective strategies by creating additional mechanisms, like the C4 cycle, which ensures a CO2-rich environment around the RubisCO, more favourable to carboxylation and thus minimizing the oxygenase activity that inhibits photosynthesis.
Notes and References
We thank Editions Dunod and QUAE for having authorized us to reproduce figures for this article.
Cover image. [Source: Photo © Jean-François Morot-Gaudry]
[1] Carbon 11 (11C) is an isotope of carbon with a half-life of 20.38 minutes. Experiments using this radioactive isotope must therefore be very short because it can no longer be detected after a few hours. It’s commonly used to mark molecules in ” positron emission tomography ».
[2] Benson, A.A. (1951) Identification of ribulose in 14CO2 photosynthesis products. J. Am. Chem. Soc. 73:2971-2972.
[3] Bassham, J. A., Benson, A. A., Kay, L. D., Harris, A. Z., Wilson, A. T. & Calvin, M. (1954) The path of carbon in photosynthesis. XXI. The cyclic regeneration of carbon dioxide acceptor. J. Am. Chem. Soc. 76:1760-1770;
[4] Biology is based on the chemistry of carbon, because of its electrochemical potential, i.e. the part of chemistry that studies the reciprocal transformations of chemical energy and electrical energy. This very high potential proves capable of maintaining four different chemical bonds at the same time, which makes it possible to multiply the various possibilities of atomic and molecular combinations, sources of the diversification of organic molecules essential to the various processes of evolution and development of life.
[5] Dalton is a standard unit of measurement, used to express the mass of atoms and molecules. Initially defined as 1/12 of the mass of a carbon 12 atom. The kilodalton (kDa) is much more used in biology and biochemistry because of the size of the molecules. Most cellular molecules typically have a mass between 20 and 100 kDa.
[6] Nitrogen is a major component of amino acids and proteins (about 6% of the mass of a protein).
[7] Otto Heinrich Warburg (1883-1970), German physician, physiologist and biochemist. Winner of the 1931 Nobel Prize in Physiology or Medicine “for his discovery of the nature and mode of action of the respiratory enzyme.”
[8] Currently, the level of CO2 in the atmosphere has exceeded 400 ppm (0.04%). Values above 415 ppm were recorded throughout 2019 at the Mauna Loa Observatory in Hawaii.
[9] Lorimer G.H. (1981). The carboxylation and oxygenation of ribulose 1,5-bisphosphate: The primary events in photosynthesis and photorespiration. Annu. Rev. Plant Physiol. 32: 349-383.
[10] Tolbert N.D. (1997). The C2 oxidative photosynthetic carbon cycle. Annu. Rev. Plant Physiol. Plant Mol. Biol. 48: 1-25.
[11] Hagemann M., Kern R., Maurino V.G., Hanson D.T., Weber A.P.M., Sage R.F. & Bauwe H. (2016) Evolution of photorespiration from cyanobacteria to land plants, considering protein phylogenies and acquisition of carbon concentrating mechanisms. J. Exp. Bot., 67:2963-2976.
[12] Erb T.J. & Zarzycki J. (2018) A short history of RubisCO: the rise and fall (?) of Nature’s predominant CO2 fixing enzyme. Curr. Opinions. Biotechnology, 49:100-107
[13] Badger M.R., Price, G.D., Long B.M. & Woodger F.J. (2006). The environmental plasticity and ecological genomics of the cyanobacterial CO2 concentrating mechanism. J. Exp. Bot. 57: 249-265.
[14] Oxaloacetate is rapidly reduced to malate which migrates into the chloroplasts of the perivascular sheath.
[15] Christin P.A. & Osborne C.P. (2014) The evolutionary ecology of C4 plants. New Phytol. 204(4):765-781; Hatch M.D. & Slack C.R. (1970). Photosynthetic CO2-fixation pathways. Ann. Rev. Plant Physiol. 21: 141-162; https://inee.cnrs.fr/fr/cnrsinfo/des-echantillons-dherbiers-revelent-les-origines-de-la-photosynthese-du-mais.
[16] Koteyeva N.K., Voznesenskaya E.V., BerryJ.O., Asaph B., Cousins A.B. & Edwards G.E. (2016) Synthesis along longitudinal leaf gradients in Bienertia sinuspersici and Suaeda aralocaspica (Chenopodiaceae). J. Exp. Bot. 67 (9): 2587-2601.
The Encyclopedia of the Environment by the Association des Encyclopédies de l'Environnement et de l'Énergie (www.a3e.fr), contractually linked to the University of Grenoble Alpes and Grenoble INP, and sponsored by the French Academy of Sciences.
To cite this article: MOROT-GAUDRY Jean-François, JOYARD Jacques (January 5, 2025), The path of carbon in photosynthesis, Encyclopedia of the Environment, Accessed May 29, 2026 [online ISSN 2555-0950] url : https://www.encyclopedie-environnement.org/en/life/path-carbon-photosynthesis-2/.
The articles in the Encyclopedia of the Environment are made available under the terms of the Creative Commons BY-NC-SA license, which authorizes reproduction subject to: citing the source, not making commercial use of them, sharing identical initial conditions, reproducing at each reuse or distribution the mention of this Creative Commons BY-NC-SA license.







